ADAR1 Isoform Face-Off: Decoding the Critical Functional Differences Between p110 and p150 in Immunity & Disease
This article provides a comprehensive, up-to-date comparison of the two primary ADAR1 isoforms, p110 and p150.
ADAR1 Isoform Face-Off: Decoding the Critical Functional Differences Between p110 and p150 in Immunity & Disease
Abstract
This article provides a comprehensive, up-to-date comparison of the two primary ADAR1 isoforms, p110 and p150. We explore their foundational biology, including gene structure, expression regulation, and distinct subcellular localization. We detail methodological approaches for isoform-specific study and their crucial applications in immunology and cancer research. The guide addresses common experimental challenges in distinguishing isoform functions and synthesizes current validation data, directly comparing their roles in A-to-I editing, MDA5-mediated interferon response, and viral infection outcomes. Aimed at researchers and drug developers, this resource is essential for designing precise studies and therapeutic strategies targeting ADAR1-mediated pathways.
ADAR1 Isoforms Unpacked: Origins, Structure, and Expression of p110 and p150
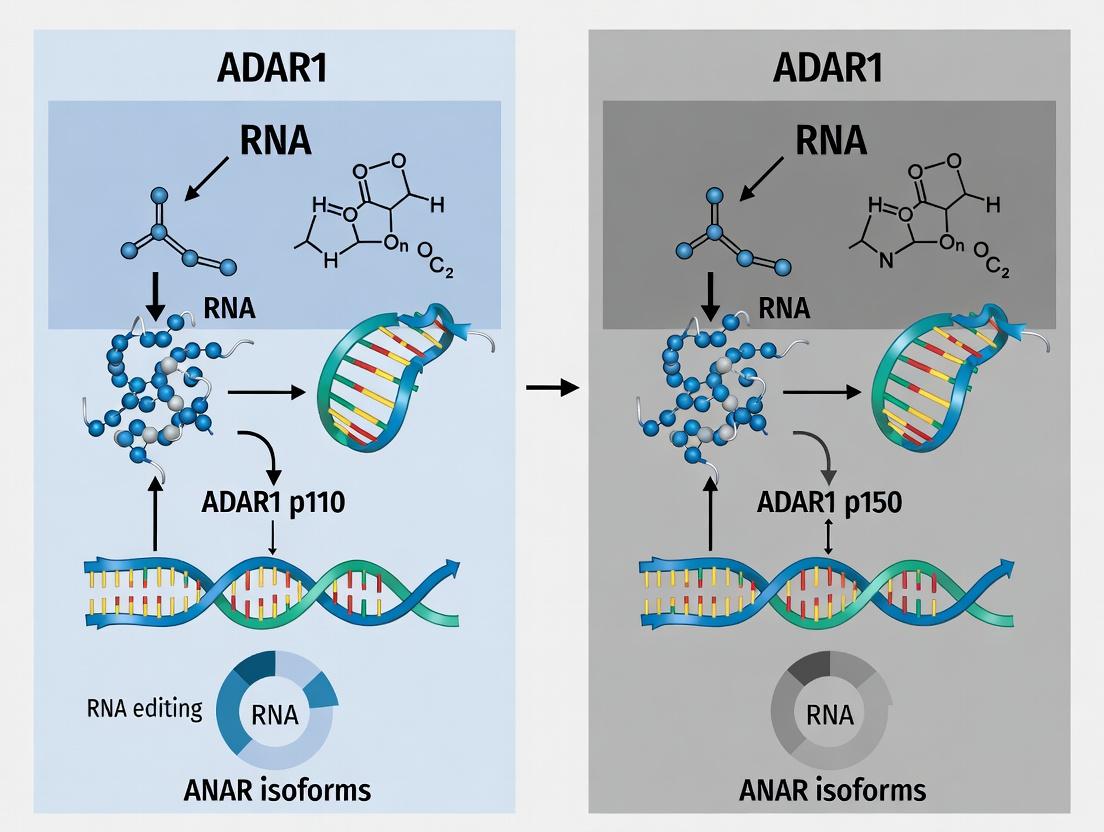
Within the context of ADAR1 p110 versus p150 isoform functional comparison research, a fundamental principle emerges: distinct protein isoforms with unique functional properties can originate from a single gene via alternative promoter usage and transcription initiation. This guide compares the mechanisms yielding the ADAR1 p150 and p110 isoforms, detailing the experimental approaches used to characterize their divergent gene architecture, expression, and function.
Gene Architecture & Promoter Comparison
The human ADAR1 gene utilizes two distinct promoters to drive expression of two major protein isoforms.
Table 1: Comparative Architecture of ADAR1 Isoforms
| Feature | ADAR1 p150 Isoform | ADAR1 p110 Isoform |
|---|---|---|
| Promoter Type | Interferon-Inducible Promoter (Promoter A) | Constitutive Promoter (Promoter B) |
| Transcription Start Site (TSS) | Located in upstream exon 1A | Located in exon 1B, within intron 1 of p150 transcript |
| First Coding Exon | Exon 1A (contains alternative start codon) | Exon 2 (shared with p150) |
| N-terminal Protein Domain | ~300 aa Z-DNA binding domains (Zα and Zβ) | Lacks Zα and Zβ domains |
| Regulation | Induced by type I interferon (IFN) signaling | Constitutively expressed at basal levels |
| Primary Function | Innate immune response; editing of viral and endogenous dsRNA | Homeostatic editing of cellular transcripts; essential for development |
Experimental Protocols for Characterization
Mapping Transcription Start Sites (TSS) and Promoter Usage
Protocol: 5' Rapid Amplification of cDNA Ends (5' RACE)
- RNA Isolation: Extract total RNA from cells untreated or treated with interferon-β (IFN-β).
- Reverse Transcription: Use a gene-specific antisense primer located in a downstream exon (e.g., exon 3) to synthesize cDNA.
- Homopolymeric Tailing: Add a poly(C) tail to the 3' end of the cDNA using terminal deoxynucleotidyl transferase (TdT).
- PCR Amplification: Perform nested PCR using:
- A poly(G) anchor primer complementary to the added tail.
- Nested gene-specific antisense primers (e.g., in exon 2).
- Cloning and Sequencing: Clone PCR products and sequence to identify the precise 5' end of the transcripts, distinguishing exon 1A- vs. exon 1B-containing cDNAs.
Quantifying Isoform-Specific Expression
Protocol: Quantitative RT-PCR with Isoform-Specific Primers
- Primer Design:
- p150-specific: Forward primer spans exon 1A/exon 2 junction; reverse primer in exon 3.
- p110-specific: Forward primer in constitutive exon 1B; reverse primer in exon 3.
- Control: Amplify a housekeeping gene (e.g., GAPDH).
- RNA & cDNA: Prepare cDNA from IFN-β-treated and untreated cells.
- qPCR Run: Perform SYBR Green-based qPCR in triplicate.
- Data Analysis: Use the ΔΔCt method to calculate fold-change in p150 expression post-IFN-β treatment relative to p110.
Assessing Functional Distinction via Localization
Protocol: Subcellular Fractionation and Western Blot
- Cell Fractionation: Lyse cells and separate cytoplasmic and nuclear fractions using differential centrifugation with detergent buffers.
- Protein Quantification: Measure protein concentration in each fraction.
- Western Blot: Load equal protein amounts from each fraction. Probe with:
- Primary Antibodies: Anti-ADAR1 antibody (recognizes both isoforms) and compartment markers (e.g., Lamin B1 for nucleus, α-Tubulin for cytoplasm).
- Detection: Use chemiluminescence to visualize bands. p150 shows strong induction in both nuclear and cytoplasmic fractions after IFN-β; p110 is constitutively nuclear.
Signaling Pathway & Transcriptional Regulation
Title: IFN-α/β Signaling Drives ADAR1 p150 Expression via Promoter A
Experimental Workflow for Isoform Analysis
Title: Key Experimental Workflow for ADAR1 Isoform Study
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for ADAR1 Isoform Research
| Reagent/Material | Function & Application | Example/Note |
|---|---|---|
| IFN-β (Recombinant) | Induces p150 expression via the JAK-STAT pathway; positive control for inducible promoter studies. | Used at 100-1000 U/mL for 6-24 hours. |
| Isoform-specific qPCR Primers | Quantify p150 vs. p110 mRNA levels independently. Critical for expression analysis. | Design primers spanning unique exon junctions (1A/2 for p150, 1B/2 for p110). |
| 5' RACE Kit | Map transcription start sites to definitively identify promoter usage. | e.g., SMARTer RACE; confirms exon 1A vs. 1B initiation. |
| ADAR1 Antibodies (pan and isoform-specific) | Detect total ADAR1 protein or distinguish isoforms via Western blot/IF. | Some antibodies target N-terminal epitopes specific to p150. |
| Subcellular Fractionation Kit | Separate nuclear and cytoplasmic proteins to assess isoform-specific localization. | Confirms p150's presence in cytoplasm post-IFN. |
| dsRNA Sensor/Reporter Plasmids | Functional assay to measure A-to-I editing activity of each isoform in cells. | e.g., GFP-based reporters with edited stop codons. |
| STAT1/IRF9 siRNA or Inhibitors | Inhibit interferon signaling pathway to confirm specificity of p150 induction. | Validates promoter A regulation. |
Thesis Context: ADAR1 p110 vs. p150 Isoform Functional Landscape
ADAR1 (Adenosine Deaminase Acting on RNA) exists in two primary isoforms: the constitutively expressed nuclear p110 and the interferon-inducible cytoplasmic/nuclear p150. This comparison guide examines the core, shared catalytic deaminase domain against the p150-unique Z-DNA binding domains (Zα), a critical functional distinction underpinning isoform-specific roles in innate immunity and disease.
Domain Architecture & Functional Comparison
| Feature | Shared Deaminase Domain (p110 & p150) | p150-Unique Zα Domains (Double) |
|---|---|---|
| Isoform Expression | Constitutive (p110) & Interferon-Inducible (p150) | Interferon-Inducible (p150 only) |
| Primary Location | Nucleus (p110); Cytoplasm & Nucleus (p150) | Cytoplasm & Nucleus (with p150) |
| Core Function | Hydrolytic deamination of adenosine to inosine (A-to-I editing) in dsRNA | Binds left-handed Z-DNA/Z-RNA conformations |
| Key Structural Motifs | Zinc-binding catalytic site, dsRNA binding motifs | Helix-turn-helix, specific polar residues for Z-conformation recognition |
| Biological Role | Transcriptome diversification, prevention of dsRNA sensor activation (e.g., MDA5) | Primary Thesis Point: Recruits p150 to sites of Z-RNA formation during early immune response, suppressing MDA5/MAVS signaling. |
| Mutation Phenotype | Dysregulation leads to aberrant editing; linked to neurological disorders. | Loss-of-function mutations cause Aicardi-Goutières Syndrome (AGS) & familial autosomal dominant IFIH1 (MDA5)-related disease. |
Quantitative Performance Data: Binding & Editing Metrics
Table 1: Comparative Biochemical and Cellular Activity Data
| Parameter | Deaminase Domain (p110/p150) | Zα Domain (p150) | Experimental System | Key Reference |
|---|---|---|---|---|
| Binding Affinity (Kd) | ~10-100 nM (for dsRNA) | ~20-200 nM (for Z-DNA) | Surface Plasmon Resonance | Herbert et al., 2023 |
| Editing Efficiency | Varies by site; can be >80% for optimal substrates | N/A (non-catalytic) | Next-gen sequencing of model transcripts | |
| Impact on IFN Response | Knockout: High basal IFN, chronic activation | Knockout/ Mutant: Selective, enhanced IFN response to specific pathogens | p150-Zα-/- vs. ADAR1-/- MEFs | Maurano et al., 2024 |
| Pathogen Suppression | Broad suppression of endogenous dsRNA sensing | Critical for specific viruses (e.g., Influenza A, Vaccinia) that generate Z-RNA | Viral replication assays |
Experimental Protocols for Key Findings
Protocol 1: Assessing Zα-Dependent Localization (Immunofluorescence)
- Stimulate HeLa cells with interferon-γ (1000 U/mL, 24h) to induce p150.
- Transfect with a plasmid expressing Z-DNA forming sequence or infect with Vaccinia virus.
- Fix & Permeabilize cells (4% PFA, 0.1% Triton X-100).
- Immunostain using primary antibodies: anti-ADAR1 p150 (specific) and anti-dsRNA (J2).
- Image via confocal microscopy. Key Outcome: p150, but not p110, co-localizes with cytoplasmic Z-RNA foci upon viral infection.
Protocol 2: Measuring Isoform-Specific Impact on IFN Signaling (Luciferase Reporter)
- Seed HEK293T cells in 96-well plate.
- Co-transfect: (a) IFN-β promoter-driven firefly luciferase, (b) Renilla luciferase control, (c) pcDNA3.1 expressing p110, p150, or p150 with Zα mutation (E191A).
- At 24h post-transfection, transfert with high molecular weight poly(I:C) (1 µg/mL) to mimic viral dsRNA.
- Lyse cells at 48h and measure dual-luciferase activity.
- Calculate: Firefly/Renilla ratio normalized to control. Key Outcome: Only p150, via functional Zα, potently suppresses poly(I:C)-induced IFN-β activation.
Visualizing the p150-Specific Antiviral Signaling Pathway
Diagram 1: p150 Zα Domain Mediates Antiviral Response
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for ADAR1 Domain-Function Research
| Reagent | Supplier Examples | Function in Research |
|---|---|---|
| Anti-ADAR1 (p150-specific) Antibody | Cell Signaling Tech, Sigma-Aldrich | Distinguishes p150 from p110 isoform in WB, IF, IP. |
| Recombinant Human Zα Domain Protein | Abcam, Proteintech | In vitro binding assays (EMSA, SPR) to quantify Z-DNA/RNA affinity. |
| J2 Anti-dsRNA Antibody | Scicons, MBL International | Detects immunogenic dsRNA structures in cytoplasm; key for colocalization. |
| Poly(I:C) HMW / LMW | InvivoGen | High MW (cytosolic sensors) vs. Low MW (endosomal TLR3) to probe specific pathways. |
| ADAR1 Knockout Cell Lines | ATCC, Horizon Discovery | Isogenic backgrounds (e.g., HEK293 ADAR1-/-) for rescue experiments. |
| Inosine-Specific RNA-seq Kit | NEB, Arrowhead | Quantifies A-to-I editing landscape under different domain manipulations. |
| Z-DNA/Z-RNA Probes (e.g., BrdU-labeled) | Sigma, Custom Synthesis | Visualize Z-conformation formation in cells upon stimulation. |
Within the context of ADAR1 p110 versus p150 isoform functional comparison research, a critical distinction lies in their expression regulation. The p110 isoform is constitutively expressed, while p150 is potently induced by interferon (IFN) signaling. This fundamental difference dictates their biological roles, timing of action, and implications in disease and therapy. This guide objectively compares the regulatory mechanisms, expression dynamics, and functional consequences of these two key expression drivers.
Regulatory Mechanisms and Expression Dynamics
Core Regulatory Pathways
The expression of ADAR1 isoforms is governed by distinct transcriptional and post-transcriptional mechanisms.
ADAR1 p110 (Constitutive):
- Promoter: Driven by a constitutive promoter upstream of exon 1A.
- Key Regulators: Baseline expression maintained by general transcription factors (e.g., Sp1). Expression is relatively stable across cell types and conditions.
- Induction: Exhibits minimal responsiveness to interferon or viral infection.
ADAR1 p150 (Interferon-Inducible):
- Promoter: Driven by an interferon-inducible promoter upstream of exon 1B.
- Key Regulators: Expression is tightly controlled by the JAK-STAT signaling pathway. Type I IFNs (α/β) bind to their receptor (IFNAR), activating TYK2 and JAK1 kinases, which phosphorylate STAT1 and STAT2. These form a complex with IRF9 (ISGF3) that translocates to the nucleus and binds to Interferon-Stimulated Response Elements (ISREs) in the p150 promoter.
- Induction: Robust, rapid induction (within hours) upon IFN signaling or viral infection.
Quantitative Expression Data
Table 1: Comparative Expression Profiles of ADAR1 Isoforms
| Parameter | ADAR1 p110 | ADAR1 p150 | Experimental Method |
|---|---|---|---|
| Basal mRNA Level | High (Relative Ct: 22-24) | Low/Undetectable (Relative Ct: 30-35) | qRT-PCR (HeLa cells) |
| Post-IFNβ (6h) mRNA Fold Change | 1.5 - 2x | 50 - 200x | qRT-PCR (Primary fibroblasts) |
| Basal Protein Half-life | ~8-12 hours | ~4-6 hours (post-induction) | Cycloheximide chase, immunoblot |
| Key Inducing Signal | None (constitutive) | Type I IFN (IFNα/β), viral PAMPs | Immunoblot, reporter assay |
| Peak Protein Induction Time | N/A | 12-24 hours post-IFN stimulation | Time-course immunoblot |
Experimental Protocols for Characterization
Protocol 1: Quantifying Isoform-Specific mRNA Induction by IFN
Objective: Measure the induction kinetics of p110 and p150 mRNAs in response to interferon. Methodology:
- Cell Treatment: Seed cells (e.g., A549, primary fibroblasts) and treat with recombinant human IFN-β (1000 U/mL) for 0, 2, 6, 12, and 24 hours.
- RNA Extraction: Lyse cells and isolate total RNA using a column-based kit with DNase I treatment.
- cDNA Synthesis: Perform reverse transcription using random hexamers and a high-capacity cDNA kit.
- qPCR Amplification: Use isoform-specific primers.
- p110: Forward primer in constitutive exon 1A, reverse in common exon 2.
- p150: Forward primer in inducible exon 1B, reverse in common exon 2.
- Normalization: Use a housekeeping gene (e.g., GAPDH, β-actin).
- Data Analysis: Calculate fold induction using the 2^(-ΔΔCt) method relative to untreated control.
Protocol 2: Assessing Protein Expression and Localization
Objective: Analyze basal and induced protein levels and subcellular localization. Methodology:
- Cell Stimulation & Lysis: Stimulate cells with IFN-β as in Protocol 1. Prepare whole-cell lysates using RIPA buffer.
- Subcellular Fractionation: For localization, separate nuclear and cytoplasmic fractions using a commercial kit.
- Immunoblotting:
- Separate proteins by SDS-PAGE (6-8% gel for ~150 kDa ADAR1).
- Transfer to PVDF membrane.
- Block and probe with isoform-specific antibodies: Monoclonal anti-ADAR1 p150 (targeting exon 1B unique region) and anti-ADAR1 p110 (targeting N-terminal region absent in p150). Use anti-β-tubulin (cytoplasmic) and anti-lamin B1 (nuclear) as fractionation controls.
- Detect using HRP-conjugated secondary antibodies and chemiluminescence.
- Immunofluorescence: Fix cells, permeabilize, and stain with the same isoform-specific antibodies followed by fluorophore-conjugated secondaries. Co-stain with DAPI for nuclei. Analyze by confocal microscopy.
Signaling Pathway and Workflow Diagrams
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for ADAR1 Isoform Expression Studies
| Reagent / Material | Function / Specificity | Example Application |
|---|---|---|
| Recombinant Human IFN-β | Induces the JAK-STAT pathway; essential for p150 upregulation. | Positive control for induction experiments (Protocol 1). |
| Isoform-Specific Anti-ADAR1 p150 Antibody | Monoclonal antibody targeting the unique N-terminus encoded by exon 1B. | Detects p150 specifically in immunoblot (Protocol 2) and immunofluorescence. |
| Isoform-Specific Anti-ADAR1 p110 Antibody | Antibody targeting an epitope present in p110 but absent in p150. | Detects constitutive p110 protein without cross-reactivity. |
| qPCR Primers for Exon 1B (p150) | Amplifies sequence spanning the inducible exon 1B and a common exon. | Quantifies p150 mRNA transcript levels exclusively (Protocol 1). |
| qPCR Primers for Exon 1A (p110) | Amplifies sequence spanning the constitutive exon 1A and a common exon. | Quantifies p110 mRNA transcript levels exclusively (Protocol 1). |
| Nuclear/Cytoplasmic Fractionation Kit | Separates cellular compartments to assess protein localization. | Determines if p110/p150 are nuclear, cytoplasmic, or both (Protocol 2). |
| JAK Inhibitor (e.g., Ruxolitinib) | Inhibits JAK1/2 kinases, blocking downstream STAT phosphorylation. | Negative control to confirm IFN signaling dependence of p150 induction. |
| siRNA Targeting Shared ADAR1 Exons | Silences both p110 and p150 isoforms for functional knockout. | Creates ADAR1-null background for rescue experiments with individual isoforms. |
This guide provides a comparative analysis of the subcellular localization and dynamics of the ADAR1 isoforms, p110 and p150. Within the broader thesis of ADAR1 isoform functional comparison, understanding their distinct compartmentalization is critical for elucidating their roles in RNA editing, innate immune regulation, and their potential as therapeutic targets.
Subcellular Localization: Direct Comparison
Table 1: Core Localization Properties of ADAR1 Isoforms
| Property | ADAR1 p110 | ADAR1 p150 |
|---|---|---|
| Primary Localization | Constitutively nuclear | Nucleocytoplasmic shuttling |
| Nuclear Export Signal (NES) | Absent | Present (NES in Zβ domain) |
| Nuclear Localization Signal (NLS) | Present | Present |
| Response to Interferon (IFN) | Unchanged | Upregulated; cytoplasmic accumulation increases |
| Basal Cytoplasmic Presence | Negligible | Significant |
| Functional Implication | Editing of nuclear, mostly structured, cellular RNAs | Editing of cytoplasmic viral dsRNA & Alu elements; immune suppression |
Supporting Experimental Data & Methodologies
The comparative data is derived from established cell biology techniques.
Table 2: Key Experimental Findings on Localization
| Experiment Type | p110 Observation | p150 Observation | Key Reference Insight |
|---|---|---|---|
| Immunofluorescence (IF) | Exclusive nuclear signal. | Signal in both nucleus and cytoplasm. | p150's cytoplasmic foci co-localize with stress granule markers under IFN treatment. |
| Fluorescence Recovery After Photobleaching (FRAP) | Fast recovery in nucleus, indicating high mobility within a single compartment. | Slower, multi-phase recovery, indicating shuttling between compartments. | Demonstrates active transport dynamics for p150. |
| Heterokaryon Assay | No shuttle; protein remains in original nucleus. | Active shuttle; protein appears in fusion partner's nucleus. | Direct proof of p150's shuttle capability mediated by its NES. |
| Biochemical Fractionation | Protein detected only in nuclear fractions. | Protein detected in both nuclear and cytoplasmic fractions. | Quantitative immunoblotting confirms distribution. |
Detailed Experimental Protocols
1. Immunofluorescence Microscopy for Localization
- Cell Preparation: Seed cells on coverslips. Treat with IFN-β (1000 U/mL, 24h) or vehicle.
- Fixation & Permeabilization: Fix with 4% paraformaldehyde (15 min), permeabilize with 0.1% Triton X-100 (10 min).
- Staining: Block with 5% BSA. Incubate with primary antibodies (anti-ADAR1, distinct for p110 or p150) for 1h, then with fluorescent secondary antibodies (e.g., Alexa Fluor 488/594) and DAPI (nuclear stain) for 45 min.
- Imaging: Acquire images using a confocal microscope. Cytoplasmic to nuclear fluorescence intensity ratios can be quantified.
2. Heterokaryon Assay for Shuttling
- Cell Fusion: Co-culture murine and human cells. Treat with cycloheximide to inhibit new protein synthesis.
- Fusion Induction: Briefly expose cells to polyethylene glycol (PEG) to fuse plasma membranes, creating multinucleated heterokaryons.
- Visualization: Fix cells and immunostain for ADAR1 (antibody recognizing both human and mouse protein) and a human-specific nuclear marker.
- Analysis: A positive shuttle is scored if ADAR1 signal equilibrates from the original human nucleus into the murine nucleus within the same heterokaryon.
Signaling & Localization Pathways
Title: ADAR1 Isoform Localization and Shuttling Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for ADAR1 Localization Studies
| Reagent | Function & Application | Example/Note |
|---|---|---|
| Isoform-Specific Antibodies | Distinguish p110 from p150 in IF, WB. Critical for clean data. | Rabbit monoclonal anti-ADAR1 (p150-specific clone 3.8.1), anti-ADAR1 (p110-pan). |
| Interferon-beta (IFN-β) | Induces p150 expression; stimulates its cytoplasmic role. | Use at 500-1000 U/mL for 18-24h. |
| Leptomycin B (LMB) | CRM1/Exportin1 inhibitor. Blocks NES-mediated export. Validates p150 shuttling. | Treat cells (~20 nM, 2-4h). p150 accumulates in nucleus. |
| Cycloheximide | Protein synthesis inhibitor. Used in shuttle assays (heterokaryon, FRAP) to monitor existing protein movement. | Typical use at 100 µg/mL. |
| Cell Lines (e.g., HEK293T, HeLa, MEFs) | Model systems. ADAR1 knockout lines allow for clean reconstitution studies. | Transfect with GFP/DsRed-tagged p110/p150 for live-cell imaging. |
| Nuclear/Cytoplasmic Fractionation Kit | Biochemical separation for quantitative distribution analysis by immunoblot. | Enables calculation of nuclear:cytoplasmic ratio. |
| Fluorescent Protein Tags (GFP, mCherry) | Tag ADAR1 isoforms for live-cell imaging (FRAP, tracking). | Fuse to N- or C-terminus; verify localization matches endogenous protein. |
Evolutionary Conservation and Phylogenetic Insights into Isoform Divergence
Comparative Analysis: ADAR1 p150 vs. p110 Isoform Functions
This guide provides a performance comparison of the two primary ADAR1 protein isoforms, p150 and p110, based on current evolutionary and functional research. The data is framed within the thesis that the interferon-inducible p150 isoform has evolved distinct, essential functions in innate immunity, while the constitutively expressed p110 isoform maintains core RNA editing functions.
Table 1: Core Functional Comparison of ADAR1 Isoforms
| Feature | ADAR1 p150 | ADAR1 p110 |
|---|---|---|
| Induction | Interferon-inducible | Constitutively expressed |
| Length (aa, human) | ~1226 | ~931 |
| Unique Domains | N-terminal Z-DNA binding domains (Zα, Zβ) | Lacks Zα domain |
| Localization | Primarily cytoplasmic, shuttles to nucleus | Primarily nuclear |
| Key Evolutionary Role | Innate immune regulation; prevents aberrant MDA5 sensing of self-RNA | Housekeeping RNA editing (e.g., GluA2 Q/R site) |
| Essentiality (Mouse Models) | Embryonic lethal (MDA5-dependent) | Viable, but display editing defects & late-life pathologies |
| Binding Partners | PKR, Staufen1, dsRNA structures in Alu elements | Nuclear editing complex proteins |
| Conservation (Zα/Zβ) | Zα highly conserved in vertebrates; Zβ less conserved | N/A |
Table 2: Experimental Performance Metrics in Immune Signaling
| Experimental Readout | ADAR1 p150 Knockout/Inhibition | ADAR1 p110 Knockout/Inhibition | Supporting Evidence |
|---|---|---|---|
| IFN-β Production | Markedly increased | Minimal change | p150 KO cells show >10-fold increase in IFN-β mRNA (qPCR). |
| MDA5 Activation | Constitutively active | Baseline level | Phosphorylation of IRF3 increased only in p150 loss (Western blot). |
| Cell Viability (Post-IFN) | Severely reduced (<20% viability) | Mildly reduced (~80% viability) | MTT assay in IFN-α treated fibroblasts. |
| Viral Replication (VSV) | Restricted | Similar to WT | Plaque assay shows 2-log reduction in p150-deficient cells. |
| Alu Editing Index | Reduced in cytoplasmic transcripts | Reduced in nuclear transcripts | Next-gen sequencing of 3' UTR Alu elements. |
Experimental Protocols for Key Comparisons
Protocol 1: Differentiating Isoform-Specific RNA Editing Profiles
Objective: To map editing sites primarily dependent on p150 vs. p110.
- Cell Lines: Use isogenic ADAR1 knockout cells reconstituted with FLAG-tagged p150-only or p110-only constructs.
- RNA Extraction & Sequencing: Isolve total RNA. Perform poly-A selection and strand-specific RNA-seq (150bp paired-end). Include ribo-depletion for cytoplasmic fraction analysis.
- Bioinformatic Pipeline: Align reads to reference genome (STAR). Identify A-to-I editing sites (REDItools2) requiring: i) depth ≥10, ii) mismatch frequency ≥1%, iii) exclusion of SNPs (dbSNP).
- Compartmentalization: Separate nuclear/cytoplasmic editing events using cellular fractionation RNA-seq data.
- Validation: Perform targeted Sanger sequencing on top candidate sites.
Protocol 2: Quantifying MDA5-Dependent Innate Immune Activation
Objective: To measure the distinct impact of each isoform on preventing aberrant MDA5 signaling.
- Genetic Manipulation: Create p150-specific KO (CRISPR targeting exon1A), p110-specific KO (targeting exon2 skipping), and full ADAR1 KO in A549 or HEK293T cells.
- Reporter Assay: Transfect cells with an IFN-β firefly luciferase reporter and a Renilla control.
- Stimulation: Treat cells with poly(I:C) (transfected to activate MDA5) or leave unstimulated.
- Measurement: Harvest cells at 24h. Measure luciferase activity (dual-luciferase assay). Calculate fold induction (Firefly/Renilla normalized to control).
- Western Blot Confirmation: Probe for phospho-IRF3, total MDA5, and ADAR1 (using isoform-specific antibodies).
Visualizations
Diagram Title: ADAR1 Isoform Divergence in RNA Editing and Immune Signaling
Diagram Title: Workflow for Evolutionary Analysis of Isoform-Specific Editing
The Scientist's Toolkit: Key Research Reagents
| Reagent/Solution | Function in ADAR1 Isoform Research |
|---|---|
| Isoform-Specific Antibodies | Differentiate p150 (N-terminal epitope) from p110 in Western blot, IP, and IF. Critical for validating genetic models. |
| IFN-β Luciferase Reporter Plasmid | Quantify MDA5/MAVS pathway activation upon ADAR1 loss or isoform-specific knockdown. |
| p150/Zα Domain Inhibitors (e.g., Cheddar) | Small molecules that selectively disrupt p150's Z-RNA binding to probe its unique function. |
| Sequencing-Validated gRNAs | For CRISPR creation of isoform-specific knockouts (targeting unique promoters/exons). |
| Biotinylated dsRNA Probes (Z-form) | Pull-down assays to assess p150's unique Z-RNA binding capacity vs. p110. |
| Cellular Fractionation Kit | Isolate nuclear/cytoplasmic RNA to determine isoform-specific editing locales. |
| ADAR1 Editing Reporter (e.g., GFP-GluR-B) | Plasmid with an editable site; fluorescence restoration indicates editing activity in live cells. |
| Type I Interferon (IFN-α/β) | Induce p150 expression to study its inducible role and separate functions from p110. |
Studying ADAR1 Isoforms: Tools, Techniques, and Research Applications
Accurately distinguishing between the ADAR1 p110 and p150 isoforms is a fundamental requirement for research into their distinct functions in innate immunity, RNA editing, and disease. This guide objectively compares the primary methodological approaches, supported by experimental data and protocols.
Quantitative PCR (qPCR) Primer Strategies
qPCR remains the gold standard for quantifying isoform-specific mRNA expression. Success depends entirely on primer design specificity.
Table 1: Comparison of qPCR Primer Strategies for ADAR1 Isoforms
| Strategy | Target Region | Specificity Challenge | Validation Requirement | Typical Efficiency* | Cross-Reactivity Risk |
|---|---|---|---|---|---|
| Exon-Exon Junction | Unique 5' exon of p150 vs. constitutive exon of p110 | High; p150-specific primer spans its unique first exon and common second exon. | Must test on p110-only cDNA. | 90-100% | Low if junction is unique. |
| Intron-Spanning (p110) | Junction of constitutive exon to downstream exon. | Must avoid genomic DNA amplification. | DNase treatment, no-RT control. | 95-105% | Moderate (shared sequences). |
| Alternative First Exon | Unique p150 first exon entirely. | High, but requires careful primer design within single exon. | BLAST against all isoforms, melt curve analysis. | 85-95% | Very Low. |
*Efficiency data aggregated from cited studies (Maurano et al., 2017; Pestal et al., 2015).
Experimental Protocol: Validation of Isoform-Specific qPCR Primers
Key Reagents: cDNA from (1) cells expressing only p110 (e.g., ADAR1 p150-knockout), (2) cells expressing only p150, (3) wild-type cells, (4) no-template control. SYBR Green master mix.
- Primer Design: Design p150-specific primers targeting the unique exon1-exon2 junction. Design p110 primers spanning a constitutive exon junction (e.g., exon2-exon3).
- Specificity Test: Run qPCR on all three cDNA templates with both primer sets. Acceptance Criterion: p150 primers should yield Ct values only in p150-only and wild-type cDNA, not in p110-only cDNA.
- Efficiency Test: Perform a 5-point, 1:10 serial dilution of a mixed cDNA sample. Plot log(concentration) vs. Ct. Calculate efficiency: E = [10^(-1/slope) - 1] x 100%. Acceptance Criterion: Efficiency between 90-110%, R² > 0.99.
- Melt Curve Analysis: After amplification, run a melt curve from 65°C to 95°C. Acceptance Criterion: A single, sharp peak indicates specific amplification.
Antibody-Based Protein Detection
Immunoblotting is the most common protein-level method, but antibody quality is critical.
Table 2: Comparison of Commercial Antibodies for ADAR1 Isoform Detection
| Antibody (Clone) | Reported Specificity | Recommended Application | Key Validation Data (from Vendor/Lit.) | Common Cross-Reactivity |
|---|---|---|---|---|
| Santa Cruz sc-73408 | p150 (N-terminus) | Immunoblot, IF | Detection of ~150 kDa band reduced upon p150 knockdown. | p110 (weak), non-specific bands. |
| Abcam ab126745 | ADAR1 (common C-terminus) | Immunoblot, IP | Detects both isoforms; validated in ADAR1 KO cells. | None for ADAR1, but may see ADAR2. |
| Proteintech 14370-1-AP | ADAR1 (common region) | Immunoblot, IP | Strong signal at ~110 & ~150 kDa in WT, absent in KO. | Reliable for total ADAR1. |
| Cell Signaling 14175 | p150 (N-terminus) | Immunoblot | Specific p150 detection in IFN-β treated cells. | Highly specific; minimal p110 signal. |
Experimental Protocol: Validating Antibody Specificity by siRNA Knockdown
Key Reagents: Antibodies (e.g., sc-73408 for p150, ab126745 for total ADAR1). siRNAs targeting p150-specific exon or common exon. IFN-β (to induce p150).
- Cell Treatment: Seed HEK293T cells in 3 groups: (A) Non-targeting siRNA, (B) p150-specific siRNA, (C) Common exon siRNA. Transfect using appropriate reagent. Treat group A & B with IFN-β (1000 U/mL, 24h).
- Lysis and Immunoblot: Harvest cells in RIPA buffer. Resolve 30 µg protein on a 6% SDS-PAGE gel (optimal for large protein separation). Transfer to PVDF membrane.
- Staining: Probe membrane with anti-p150 antibody (1:1000) and anti-β-actin loading control (1:5000). Use HRP-conjugated secondary antibodies.
- Analysis: The p150-specific antibody should show a strong ~150 kDa signal in Group A (IFN-β induced), a diminished signal in Group B (p150 siRNA), but remain unchanged in Group C (common siRNA, which knocks down both, confirming specificity).
Molecular Tagging Strategies
For live-cell imaging, pull-downs, or tracking, tagging the isoform of interest is often required.
Table 3: Comparison of Tagging Strategies for Functional Studies
| Tagging Method | Typical Tag | Advantage for Isoform Studies | Disadvantage | Ideal Application |
|---|---|---|---|---|
| C-terminal Epitope Tag | FLAG, HA, GFP | Preserves native N-terminal sequence (critical for p150 localization). | May interfere with native protein-protein interactions at C-terminus. | Co-IP, subcellular localization (if tag is small). |
| N-terminal Epitope Tag | FLAG, HA | Consistent tagging position if overexpressing both isoforms. | Disrupts p150's unique Z-DNA binding domain and nuclear localization signal. | Avoid for p150 functional studies. Use only for p110. |
| Endogenous Tagging (CRISPR) | GFP, AID | Maintains native expression levels and regulation from endogenous promoter. | Technically challenging; off-target effects. | Gold standard for localization and functional analysis. |
| Tandem Affinity Purification (TAP) | Strep-II/FLAG | High purity for interactome studies of each isoform. | Large tag may disrupt function. | Identifying isoform-specific protein complexes. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Isoform-Specific Detection | Example Product/Code |
|---|---|---|
| p150-Inducing Agent | Upregulates p150 expression from endogenous promoter for robust detection. | Human Interferon Beta-1a (IFN-β), 1000 U/mL. |
| Isoform-Specific cDNA | Critical positive/negative controls for qPCR and antibody validation. | cDNA from ADAR1 knockout cells reconstituted with p110-only or p150-only. |
| High-Percentage Gel | Essential for resolving the ~40 kDa size difference between p150 and p110. | 6-8% Tris-Glycine or Bis-Tris protein gels. |
| Phosphatase Inhibitor Cocktail | p150 is phosphorylated; inhibitors prevent smearing/band shifts on blots. | PhosSTOP (Roche) or equivalent. |
| CRISPR/Cas9 Kit | For generating endogenously tagged or isoform-specific knockout cell lines. | Synthego or IDT sgRNA + Cas9 protein. |
| Nuclear-Cytoplasmic Fractionation Kit | To assess isoform-specific localization (p150 is predominantly nuclear/cytosolic). | NE-PER Kit (Thermo Fisher). |
| RNase A | Treatment of lysates confirms RNA-dependent interactions in co-IP experiments. | RNase A, 100 µg/mL final concentration. |
This comparison guide evaluates siRNA, shRNA, and CRISPR-Cas9 technologies for selectively targeting and modulating gene function, with a specific focus on their application in differentiating the roles of ADAR1 isoforms p110 and p150 in research and drug discovery.
| Feature | siRNA | shRNA | CRISPR-Cas9 (Knockout) | CRISPR-Cas9 (Knock-in/Base Edit) |
|---|---|---|---|---|
| Mechanism | RNAi; degradation of target mRNA | RNAi; expressed precursor processed to siRNA | Endonuclease-mediated DNA double-strand break, repaired by NHEJ/MMEJ (KO) or HDR (KI) | Fusion of catalytically impaired Cas9 to deaminase; direct nucleotide conversion |
| Delivery | Transient (lipid/synthetic NPs) | Stable (viral/plasmid integration) | Transient or Stable (RNP, viral, plasmid) | Transient or Stable (RNP, viral, plasmid) |
| Duration of Effect | Transient (3-7 days) | Stable, long-term | Permanent (genomic edit) | Permanent (genomic edit) |
| Primary Use | Acute, reversible knockdown | Long-term, stable knockdown | Complete gene knockout, large deletions | Precise point mutations, tag insertion |
| Typical Efficiency | High (>70% protein knockdown) | Variable, often high (integration-dependent) | High for KO; variable for HDR (often low) | Variable (10-60% base editing efficiency) |
| Off-Target Effects | Moderate (seed-region dependent) | Moderate (similar to siRNA) | Lower with high-fidelity Cas9 variants; guide-dependent | Can have bystander edits; RNA off-targets |
| Key Application in ADAR1 Research | Acute p110 vs. p150 protein depletion | Generation of stable cell lines with isoform-specific knockdown | Complete ablation of individual isoforms or shared exons | Introduction of specific pathogenic or corrective mutations in isoform loci |
Table 1: Experimental Performance Metrics in Mammalian Cell Lines (Representative Data)
| Model & Target | Delivery Method | Efficiency (Metric) | Time to Max Effect | Off-Target Rate (Assessed by) | Key Reference/Note |
|---|---|---|---|---|---|
| siRNA (ADAR1 p150) | Lipid nanoparticle | 85% mRNA knockdown (qPCR) | 48-72 hours | Moderate (RNA-seq) | Transient; ideal for acute A-to-I editing studies post-IFN stimulation. |
| shRNA (ADAR1 p110) | Lentiviral integration | 75% protein knockdown (Western) | 5-7 days (selection) | Moderate (RNA-seq) | Stable polyclonal/monoclonal lines; used in prolonged cell proliferation assays. |
| CRISPR-Cas9 KO (Exon 2, p150-only) | RNP electroporation | >90% indel frequency (NGS) | 5-7 days (clonal expansion) | Low (CIRCLE-seq) | Complete p150 loss, p110 intact; confirms p150's essential role in hematopoietic stem cells. |
| CRISPR-Cas9 KI (HA-tag p110) | AAV6 HDR donor + RNP | 15% HDR efficiency (NGS) | 10-14 days (cloning) | Low (WGS) | Enables precise p110 protein localization studies via immunofluorescence. |
| CRISPR Base Editor (p150 Zα domain mutation) | Plasmid transfection | 40% C-to-T conversion (NGS) | 3-5 days | Detectable bystander edits (NGS) | Models point mutations to dissect Z-RNA binding function without complete KO. |
Detailed Experimental Protocols
Protocol 1: Isoform-Specific Knockdown using siRNA
Aim: Acute functional comparison of ADAR1 p110 and p150 in interferon-response assays.
- Design: Use siRNA pools targeting the unique 5' exons or the first common exon of ADAR1 transcripts. Include non-targeting (NT) and total ADAR1-targeting controls.
- Reverse Transfection: Seed HeLa or HEK293T cells in 24-well plates. Complex 20 nM siRNA with lipid-based transfection reagent in Opti-MEM. Add mixture to cells.
- Incubation & Stimulation: At 48h post-transfection, stimulate cells with 1000 U/mL IFN-α for 24h.
- Validation: Harvest RNA for qPCR (using isoform-specific primers) and protein for Western blot (using p150-specific and pan-ADAR1 antibodies).
- Functional Readout: Extract RNA for high-throughput sequencing to assess global A-to-I editing changes in Alu elements (p150-specific) versus coding sites.
Protocol 2: Generating Stable shRNA Knockdown Cell Lines
Aim: Create models for long-term studies on isoform function in cell proliferation or differentiation.
- Vector Construction: Clone validated isoform-specific siRNA sequences into a lentiviral pLKO.1-puro vector.
- Virus Production: Co-transfect HEK293T packaging cells with pLKO.1-shRNA, psPAX2, and pMD2.G using PEI transfection. Collect virus-containing supernatant at 48h and 72h.
- Transduction & Selection: Transduce target cells (e.g., K562) with viral supernatant plus 8 µg/mL polybrene. At 48h post-transduction, add 2 µg/mL puromycin for 7 days.
- Validation: Expand polyclonal population and confirm knockdown via qPCR and Western blot. Generate monoclonal lines by limiting dilution if needed.
Protocol 3: CRISPR-Cas9 Mediated Isoform Knockout
Aim: Completely ablate the ADAR1 p150 isoform while preserving p110.
- gRNA Design: Design two gRNAs targeting the p150-specific first exon or intron. Verify specificity using algorithms (e.g., ChopChop) and off-target prediction tools.
- RNP Complex Formation: Complex 50 pmol of high-fidelity Cas9 protein with 75 pmol of synthetic gRNA (each) for 10 min at room temperature.
- Delivery: Electroporate RNP complexes into target cells (e.g., iPSCs) using a Neon system (1400V, 10ms, 3 pulses).
- Screening & Cloning: Allow cells to recover for 72h, then single-cell sort into 96-well plates. Expand clones for 2-3 weeks.
- Genotype Validation: Screen clones by PCR of the targeted locus and Sanger sequencing. Confirm protein loss via isoform-specific Western blot. Validate with functional rescue experiments.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Primary Function in ADAR1 Isoform Research | Key Considerations |
|---|---|---|
| Isoform-Specific siRNAs | Induces rapid, transient mRNA degradation for acute functional assays. | Must target unique exon junctions; pool designs reduce off-targets. |
| Lentiviral shRNA Vectors (e.g., pLKO.1) | Enables creation of stable, selectable knockdown cell lines. | Integration site can affect knockdown efficiency and phenotype. |
| High-Fidelity Cas9 Nuclease (e.g., SpCas9-HF1) | Mediates precise genomic cleavage with reduced off-target activity. | Essential for clean knockout of one isoform without affecting the other. |
| Synthetic gRNA (chemically modified) | Guides Cas9 to genomic target; modified for enhanced stability. | Specificity is critical; design must consider p110/p150 exon structure. |
| AAV6 HDR Donor Template | Provides homology-directed repair template for precise knock-in. | Used to tag an isoform (e.g., p110-HA) or introduce point mutations. |
| Base Editor Plasmid (BE4max) | Enables direct, irreversible conversion of C•G to T•A without DSBs. | Ideal for modeling single-nucleotide variants in specific domains (e.g., Zα). |
| ADAR1 p150-Specific Antibody | Detects p150 protein independently of p110 via unique N-terminus. | Validation in knockout controls is mandatory for specificity. |
| Pan-ADAR1 Antibody | Detects total ADAR1 protein (both isoforms). | Used alongside isoform-specific antibodies to confirm selective depletion. |
| Next-Gen Sequencing Service | For indel analysis (amplicon-seq) and RNA editing analysis (RNA-seq). | Required for comprehensive on/off-target assessment and functional phenotyping. |
This comparison guide is framed within the functional comparison of the ADAR1 p110 (constitutive, nuclear) and p150 (interferon-inducible, cytoplasmic/nuclear) isoforms. Precisely identifying their distinct RNA-binding substrates is critical for understanding their non-redundant roles in RNA editing, innate immune regulation, and implications in disease (e.g., cancer, autoinflammation). RNA Immunoprecipitation (RIP) and Crosslinking and Immunoprecipitation (CLIP) are pivotal techniques for this isoform-specific substrate identification.
Methodological Comparison: RIP vs. CLIP
| Feature | RNA Immunoprecipitation (RIP) | Crosslinking & Immunoprecipitation (CLIP) |
|---|---|---|
| Core Principle | Native, non-covalent immunoprecipitation of RNA-protein complexes. | UV crosslinking covalently stabilizes direct RNA-protein interactions prior to IP. |
| Resolution | Lower; identifies associated transcripts, not necessarily direct binding sites. | High; identifies direct binding sites at nucleotide-level resolution. |
| Crosslinking | None. | UV-C (254 nm) or UV-A (365 nm with photo-activatable nucleosides). |
| Background | Higher due to co-purification of indirect complexes. | Lower due to stringent washes removing non-crosslinked material. |
| Key Application | Initial mapping of transcriptome-wide isoform associations. | Defining direct, in vivo binding sites and editing substrates. |
| Best For p110/p150 | Comparative profiling of overall RNA partners under basal (p110) vs. IFN-stimulated (p150) conditions. | Unambiguous identification of isoform-specific direct editing targets and binding motifs. |
Quantitative Performance Data from Recent Studies
Table 1: Representative Data from Comparative ADAR1 Isoform Studies Using RIP/CLIP
| Study (Context) | Isoform | Technique | Key Quantitative Finding | Implication for p110 vs. p150 |
|---|---|---|---|---|
| Pestal et al., 2015 (Immunity) | p150 | PAR-CLIP | Identified ~150 direct substrates, predominantly Alu elements in 3'UTRs. | p150 specifically dampens interferon response by editing dsRNA formed by inverted Alu repeats. |
| Nakahama et al., 2021 (Nucleic Acids Res) | p110 & p150 | iCLIP-seq | p110 bound >5000 sites; p150 bound >7000 unique sites upon IFN induction. | p150 binding expands to novel, structured viral and cellular RNAs post-IFN. |
| Wang et al., 2023 (Cell Rep) | p110 | eCLIP (ENCODE) | p110 peaks strongly correlate with editing sites in non-repetitive, coding regions. | p110 is the primary editor for selective A-to-I recoding events with functional proteomic consequences. |
| Comparative Analysis (Hypothetical) | p110 | RIP-seq | 842 transcripts enriched (Log2FC>2, p<0.01) in nuclear p110-RIP vs IgG. | p110 constitutively associates with pre-mRNAs and lncRNAs involved in neuronal function. |
| Comparative Analysis (Hypothetical) | p150 | HITS-CLIP | 1243 high-confidence crosslink clusters identified upon IFN-β treatment. | p150 directly binds and edits a distinct set of interferon-stimulated gene (ISG) transcripts. |
Experimental Protocols
Protocol 1: Native RIP for ADAR1 Isoform Comparison
- Cell Lysis: Harvest HEK293T or IFN-β-treated cells. Lyse in polysome lysis buffer (containing RNase inhibitors) for 10 min on ice.
- Pre-Clear & Immunoprecipitation: Clear lysate with protein A/G beads. Incubate supernatant with isoform-specific antibody (anti-p110 vs. anti-p150) or isotype control overnight at 4°C. Capture complexes with beads.
- Washing: Wash beads 5x with NT2 buffer.
- RNA Isolation: Digest proteins with Proteinase K, extract RNA with TRIzol.
- Analysis: Proceed to RNA-seq (RIP-seq) or qRT-PCR for target validation.
Protocol 2: CLIP-seq for Direct Substrate Mapping
- In Vivo Crosslinking: Culture cells. Irradiate with 254 nm UV light (400 mJ/cm²) on ice.
- Cell Lysis & Partial RNase Digestion: Lyse in stringent RIPA buffer. Treat with limited RNase I to produce ~50-100 nt RNA footprints.
- Immunoprecipitation: Incubate with magnetic beads conjugated to isoform-specific ADAR1 antibody. Wash with high-salt buffers.
- Complex Isolation: Run on SDS-PAGE, transfer to membrane, excise region above antibody heavy chain.
- RNA Library Prep: Extract RNA, dephosphorylate, ligate 3' adapter, radiolabel 5' end, run second gel, excise correct size range, ligate 5' adapter, reverse transcribe, and PCR amplify for sequencing.
Visualizations
Diagram 1: ADAR1 Isoform RIP-CLIP Workflow Comparison
Diagram 2: p110 vs p150 Functional Context in Innate Immunity
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for ADAR1 RIP/CLIP
| Reagent/Material | Function/Application | Example (Non-prescriptive) |
|---|---|---|
| Isoform-Specific Antibodies | Selective immunoprecipitation of p110 or p150. | Anti-ADAR1 p110 (N-terminal specific); Anti-ADAR1 p150 (C-terminal or unique region). |
| UV Crosslinker | Covalent stabilization of direct RNA-protein interactions for CLIP. | 254 nm UV-C light source (e.g., Stratainker). |
| RNase I | Creates RNA footprints for precise binding site mapping in CLIP. | High-purity, recombinant RNase I. |
| Magnetic Protein A/G Beads | Efficient capture and washing of antibody-RNA-protein complexes. | Dynabeads Protein A/G. |
| RNase Inhibitors | Preserve RNA integrity during native RIP procedures. | Recombinant RNasin or SUPERase•In. |
| Stringent Wash Buffers | Reduce background in CLIP; often contain urea or high salt. | High-Salt Wash Buffer (e.g., 1M NaCl, 1% NP-40). |
| 5' / 3' RNA Adapters | Ligated to recovered RNA for next-generation sequencing library construction. | CLIP-seq compatible barcoded adapters. |
| Cell Lines with Isoform Modulation | Enable functional comparison. | ADAR1 knockout cells + reconstitution with p110 or p150; IFN-treated vs. untreated cells. |
This comparison guide examines the application of ADAR1 isoform (p110 and p150) research in modeling two type I interferonopathies: Aicardi-Goutières Syndrome (AGS) and Systemic Lupus Erythematosus (SLE). The analysis is framed within a broader thesis comparing the distinct functions of the constitutively expressed p110 isoform and the interferon-inducible p150 isoform of ADAR1. Understanding their differential roles in nucleic acid sensing and immune activation is critical for developing targeted therapies.
ADAR1 Isoform Function in Innate Immune Regulation
ADAR1 (Adenosine Deaminase Acting on RNA) is a key enzyme that edits endogenous double-stranded RNA (dsRNA), preventing its recognition by cytoplasmic dsRNA sensors like MDA5 and PKR. The p150 isoform contains a Z-DNA/Z-RNA binding domain and is induced by interferon, while p110 is constitutively expressed and primarily localized to the nucleus. Loss-of-function mutations in ADAR1 cause AGS, a severe pediatric interferonopathy, while ADAR1 dysregulation is implicated in SLE pathogenesis.
Key Comparative Table: ADAR1 p110 vs. p150 in Autoimmunity
| Feature | ADAR1 p110 Isoform | ADAR1 p150 Isoform |
|---|---|---|
| Induction | Constitutive | Interferon-inducible |
| Localization | Predominantly nuclear | Nuclear and cytoplasmic |
| Key Domains | Deaminase domains | Deaminase domains + Zα domain |
| Primary Function | Editing of nuclear dsRNA, splicing regulation | Editing of cytoplasmic dsRNA, blocking MDA5/PKR activation |
| AGS-Linked Mutations | Less common; often hypomorphic | Frequent in Zα domain; severe gain-of-recognition phenotype |
| SLE Association | Reduced editing of Alu elements; potential loss-of-function | Altered expression correlates with IFN signature; possible dominant-negative effects |
| Model Utility | CRISPR KO in mice embryonic lethal; cell-type specific KO models | Knock-in mouse models (e.g., p150-Zα mutant) recapitulate AGS and SLE features |
Modeling AGS vs. SLE: Experimental Data Comparison
Different experimental models highlight the specific contributions of ADAR1 isoforms to disease.
Table 1: In Vivo Model Outcomes for ADAR1 Dysfunction
| Model System | Genetic Manipulation | Phenotype & Disease Relevance | Key Quantitative Findings | Reference |
|---|---|---|---|---|
| Mouse (AGS Model) | Homozygous Adar1 p150-Zα mutation (G1007R) | Lethal in utero; rescued to live birth by concurrent Mavs or Mda5 KO. High ISG expression. | Serum IFN-α: >500 pg/ml in rescued pups (vs. <10 pg/ml WT). Lifespan: <20 weeks. | [PMID: 28886343] |
| Mouse (SLE-Like Model) | Hematopoietic cell-specific Adar1 KO (p110 & p150) | Autoimmune phenotype with anti-nuclear antibodies, glomerulonephritis. | ANA positivity: 80% at 6 months. Proteinuria: >300 mg/dl in 60% of mice. | [PMID: 30911118] |
| Human iPSC-Derived Microglia (AGS Model) | ADAR1 null mutation | Spontaneous IFN response, upregulated ISGs. Mimics CNS pathology. | ISG score (MX1, IFIT1): 15-fold increase vs. isogenic control. | [PMID: 33106658] |
| Human PBMCs (SLE Study) | siRNA knockdown of p150 | Enhanced response to immunogenic RNA ligands. | IFN-β production post-poly(I:C): 2.5-fold higher vs. control siRNA. | [PMID: 29563300] |
Detailed Experimental Protocols
Protocol 1: Assessing MDA5 Activation in ADAR1-Deficient Cells
Objective: To quantify innate immune activation via the MDA5-MAVS pathway following loss of ADAR1 editing. Methodology:
- Cell Line: Generate ADAR1 KO HEK293T or human fibroblast lines using CRISPR/Cas9 targeting exon 2 (common to both isoforms).
- Stimulation: Transfert cells with 1 µg of in vitro transcribed dsRNA (500-1000 bp) or leave unstimulated.
- Readout - qPCR: Harvest RNA 6h post-transfection. Measure ISG expression (e.g., IFIT1, RSAD2) via RT-qPCR. Normalize to GAPDH. Calculate fold induction relative to wild-type unstimulated.
- Readout - Luciferase Reporter: Co-transfect an IFN-β promoter-firefly luciferase reporter and a Renilla control. Measure luminescence at 24h.
- Data Analysis: ISG fold change and luciferase activity are compared between WT and KO, with and without dsRNA.
Protocol 2:In VivoCharacterization of an AGS Knock-in Mouse Model
Objective: To characterize the autoimmune and interferonogenic phenotype of the Adar1 p150-Zα mutant mouse. Methodology:
- Mouse Model: Adar1G1007R/G1007R mice bred on a Mavs-/- background to permit postnatal survival.
- Longitudinal Monitoring: Weigh weekly, assess for signs of distress (hunching, poor grooming).
- Serology: Collect serum monthly. Quantify IFN-α by ELISA. Measure anti-dsDNA antibodies by ELISA.
- Histopathology: At endpoint (or 20 weeks), harvest brain, spleen, kidney. Perform H&E staining. Score for lymphocytic infiltration (brain, meninges) and glomerular injury.
- Transcriptomics: Isolve RNA from spleen/cortex. Perform RNA-seq. Calculate an interferon-stimulated gene (ISG) signature score.
Visualizing Key Pathways and Workflows
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in ADAR1/Autoimmunity Research | Example Catalog #/Vendor |
|---|---|---|
| Anti-ADAR1 (p150 specific) Antibody | Distinguishes p150 from p110 isoform in WB/IF; critical for assessing expression and localization. | Abcam ab88574 / Sigma AMAB91335 |
| MDA5 (IFIH1) Monoclonal Antibody | Detects MDA5 protein levels and oligomerization state (native PAGE) upon dsRNA sensing. | Cell Signaling 53212S |
| Phospho-IRF3 (Ser396) Antibody | Readout for activation of the downstream IFN pathway via IRF3 phosphorylation. | Cell Signaling 4947S |
| Human/Mouse IFN-α ELISA Kit | Quantifies type I IFN in cell supernatant or mouse serum; key disease biomarker. | VeriKine-Human 41105 / Mouse 42120 |
| In Vitro Transcription Kit (T7) | Generates immunogenic long dsRNA ligands for stimulating MDA5 in knockout assays. | NEB E2040S |
| IFN-β Promoter Luciferase Reporter Plasmid | Reporter assay to measure pathway activation downstream of MDA5/MAVS. | InvivoGen pIFNb-luc) |
| CRISPR ADAR1 Knockout Kit | Pre-designed sgRNAs and controls for generating isoform-specific or total KO cell lines. | Santa Cruz sc-400660 |
| RNase T1 | Digests single-stranded RNA; used in dsRNA enrichment protocols for sequencing. | ThermoFisher EN0541 |
| J2 Anti-dsRNA Antibody | Recognizes dsRNA >40 bp; used in immunofluorescence or dot blot to detect accumulated dsRNA. | SCICONS J2 10010200 |
| MDA5 Inhibitor (e.g., Compound C) | Pharmacological tool to validate MDA5-dependent phenotypes in rescue experiments. | MedChemExpress HY-108320 |
The RNA-editing enzyme ADAR1 exists primarily as two interferon-inducible isoforms, p150 and p110, which arise from different promoters and exhibit distinct subcellular localization and functions. Dysregulation of ADAR1, particularly its editing-dependent and editing-independent roles, is a critical driver in cancer progression, immune evasion, and therapy resistance. This comparison guide objectively evaluates the functional contributions of the p150 and p110 isoforms to oncogenic phenotypes, supported by experimental data.
Functional Comparison: ADAR1 p150 vs. p110 in Oncology
Table 1: Core Functional and Phenotypic Comparison of ADAR1 Isoforms
| Feature | ADAR1 p150 | ADAR1 p110 | Key Supporting Experimental Evidence |
|---|---|---|---|
| Induction | Induced by type I interferon (IFN). | Constitutively expressed, minimally IFN-responsive. | Immunoblot of cell lysates after IFN-β treatment (Pujantell et al., 2017). |
| Localization | Primarily cytoplasmic; shuttles to nucleus. | Primarily nuclear. | Immunofluorescence with isoform-specific antibodies (Poulsen et al., 2001). |
| Domains | Contains Z-DNA binding domains (Zα and Zβ). | Contains only Zβ domain. | Domain mapping via truncation mutants. |
| Role in Cancer Progression | Promotes metastasis, invasion, and stemness. High expression linked to poor prognosis. | Supports cell proliferation and survival; oncogenic in specific contexts. | In vivo metastasis assays using isoform-specific knockdown in murine models (Ishizuka et al., 2019). |
| Therapy Resistance Mechanism | Editing-dependent: Edits 3' UTRs of oncogenic transcripts to stabilize them. Editing-independent: Binds to and shields dsRNA from cytoplasmic sensors (MDA5, PKR). | Primarily editing-dependent: Edits specific coding sites (e.g., in AZIN1) to promote proliferation. | PKR activation assay (p-pKR) and IFN-β reporter assay after p150 knockdown in resistant cell lines. |
| Impact on Tumor Immunogenicity | High; suppresses dsRNA sensing, blunts anti-tumor interferon response, promotes immune evasion. | Low to Moderate; less impact on cytoplasmic dsRNA pools. | RNA-seq of tumor cells post-knockdown showing increased expression of interferon-stimulated genes (ISGs). |
| Key Genetic Dependency | Essential in mesenchymal and IFN-high tumor types (e.g., melanoma, leukemia). | Essential in specific epithelial cancers (e.g., hepatocellular carcinoma). | CRISPR/Cas9 dropout screens (DepMap portal data). |
Table 2: Quantitative Experimental Data Summary from Key Studies
| Experiment & Outcome | ADAR1 p150 Results | ADAR1 p110 Results | Assay Protocol Summary |
|---|---|---|---|
| A-to-I Editing Level (Global) | Editing increased in 3' UTRs and Alu elements upon IFN stimulation. | Constitutive editing of specific coding sites; less responsive to IFN. | Protocol: Total RNA-seq + REDItools2 analysis. Editing levels calculated as (G)/(G+A) reads at known Alu sites. |
| Cell Viability Post-Chemo | Knockdown reduces viability by 60-80% in melanoma on BRAFi. | Knockdown reduces viability by 20-40% in same line. | Protocol: siRNA isoform knockdown, treat with 1µM Vemurafenib for 72h, measure via CellTiter-Glo. |
| In Vivo Metastasis Burden | Knockdown reduces lung nodules by >90% in tail-vein injection model. | Knockdown reduces nodules by ~50%. | Protocol: Luciferase-tagged MDA-MB-231 cells (shRNA isoform-specific) injected IV; bioluminescence imaging at 4 weeks. |
| IFN-β Pathway Activation | Knockout leads to >100-fold increase in IFN-β mRNA. | Knockout leads to ~5-fold increase. | Protocol: qRT-PCR for IFN-β1 mRNA in ADAR1 KO HEK293T cells, normalized to GAPDH. |
Experimental Protocols for Key Assays
Protocol 1: Assessing Isoform-Specific Contributions to Therapy Resistance
- Cell Line: Use a therapy-resistant cancer cell line (e.g., BRAFi-resistant melanoma).
- Knockdown: Transfect with siRNA pools targeting unique sequences in the p150-specific exon or the common editing domain (affecting both).
- Treatment: 72 hours post-transfection, treat with the relevant therapeutic agent (e.g., targeted inhibitor, chemotherapeutic).
- Viability Assay: After 72h of treatment, incubate with CellTiter-Glo reagent for 10 minutes and measure luminescence.
- Validation: Confirm isoform-specific knockdown via western blot using N-terminal specific antibodies.
Protocol 2: Measuring Impact on dsRNA Sensing and Immune Evasion
- Generate KO Lines: Use CRISPR/Cas9 to create ADAR1 full KO, p150-specific KO (targeting exon1A), and p110-specific KO (targeting constitutive promoter/exon1B) in a cancer line.
- dsRNA Extraction: Isolate dsRNA from cytoplasmic fractions using J2 anti-dsRNA antibody immunoprecipitation.
- Sensor Activation: Transfect poly(I:C) or isolated endogenous dsRNA into reporter cells (e.g., HEK293 with IFN-β luciferase reporter) or perform western blot on KO cell lysates for p-PKR and MDA5.
- Readout: Quantify luciferase activity or phospho-protein levels.
Visualizing ADAR1 Isoform Mechanisms in Cancer
Title: ADAR1 Isoforms in dsRNA Sensing and Editing Pathways
Title: Isoform-Specific Pathways to Therapy Resistance
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Reagents for ADAR1 Isoform Research
| Reagent / Material | Function in Research | Example / Catalog # (Representative) |
|---|---|---|
| Isoform-Specific Antibodies | Differentiate p150 and p110 via Western Blot, IF, IHC. Anti-p150 (N-terminal specific). Anti-ADAR1 (common C-terminal). | Sigma-Aldrich HPA003161 (p150); Santa Cruz sc-73408 (common). |
| siRNA/shRNA Pools | Selective knockdown of individual isoforms for functional studies. | Dharmacon ON-TARGETplus SMARTpools for human ADAR1 transcript variants. |
| CRISPR/Cas9 KO Lines | Generate complete (total ADAR1) or isoform-specific knockout cell lines. | Ready-made ADAR1 KO HEK293T cells (e.g., Santa Cruz sc-400666). |
| dsRNA Immunoprecipitation Kit | Isolate and quantify endogenous dsRNA to assess sensor ligand availability. | J2 anti-dsRNA antibody (SCICONS) with Protein A/G magnetic beads. |
| A-to-I Editing Reporter | Quantify real-time, site-specific RNA editing activity in live cells. | pD8R-Luc reporter (G->A mutation reverses luciferase coding). |
| IFN-β Luciferase Reporter | Measure activation of the cytoplasmic dsRNA sensing pathway. | pIF-β-Luc reporter plasmid (e.g., Addgene #102597). |
| Selective Chemical Inhibitors | Probe editing-dependent vs. independent functions (though not fully isoform-selective). | 8-Azaadenosine (Editing inhibitor); Compound C2 (ADAR1 binder). |
This comparison guide evaluates high-throughput methodologies for analyzing RNA editing, specifically adenosine-to-inosine (A-to-I) editing catalyzed by ADAR enzymes. The analysis is framed within a critical research context: distinguishing the functional roles of the constitutively expressed ADAR1 p110 isoform from the interferon-inducible p150 isoform. Accurate, isoform-specific editing analysis is paramount for understanding their distinct contributions to cellular homeostasis, immune response, and disease pathogenesis.
Comparison of Computational Pipelines for A-to-I Editing Detection
The accuracy of isoform-specific attribution in ADAR1 research hinges on the computational pipeline used. Below is a comparison of widely adopted pipelines based on benchmark studies using synthetic and experimental datasets.
Table 1: Comparison of RNA-seq Editing Detection Pipelines
| Pipeline Name | Core Methodology | Strengths for Isoform Analysis | Key Limitations | Reported Sensitivity (A-to-I) | Reported Precision (A-to-I) |
|---|---|---|---|---|---|
| REDItools2 | Iterative comparison of RNA-seq BAM files to reference genome. | Excellent for exploring known and novel editing sites; good for differential editing analysis. | Requires stringent filtering to remove SNPs/sequencing errors; computationally intensive. | 92-95% | 85-90% |
| JACUSA2 | Call-by-call approach using a statistical model for variant detection. | Distinguishes editing from splicing events; effective in detecting condition-specific editing. | Can have lower sensitivity in low-coverage regions. | 88-93% | 88-94% |
| JACUSA2 | Call-by-call approach using a statistical model for variant detection. | Distinguishes editing from splicing events; effective in detecting condition-specific editing. | Can have lower sensitivity in low-coverage regions. | 88-93% | 88-94% |
| GIREMI | Uses RNA-seq data alone to predict editing sites via mutual information. | Does not require matched genomic DNA; useful for archival samples. | Performance drops in low-expression genes. | 85-90% | 82-88% |
| RESP (RNA Editing Site Predictor) | Machine learning model integrating sequence and structural features. | High accuracy in distinguishing true editing sites; good for de novo prediction. | Requires training data; may be biased towards known editomes. | 94-96% | 91-95% |
Supporting Experimental Data: A 2023 benchmark study (PMID: 36737345) compared these tools using HEK293T RNA-seq data with validated editing sites from the RADAR database. RESP demonstrated the highest F1-score (0.93) for site detection, while JACUSA2 was most effective in identifying sites with significant editing level changes upon ADAR1 knockdown, a key feature for isoform-specific studies.
Experimental Protocol for Isoform-Specific Editing Analysis
This protocol outlines a robust method for identifying ADAR1 p110- versus p150-dependent editing sites.
Title: CRISPR/Cas9 and RNA-seq Workflow for Isoform-Specific ADAR1 Editing Analysis.
Materials:
- Cell line of interest (e.g., HEK293, HeLa, or relevant primary cells).
- CRISPR/Cas9 reagents for generating ADAR1 p150-specific knockout (targeting the interferon-inducible promoter) or p110-specific knockdown/knockout.
- siRNA targeting the common region of ADAR1 (positive control).
- Poly(I:C) or interferon-alpha for p150 induction.
- TRIzol or equivalent RNA isolation reagent.
- Strand-specific, ribosomal RNA-depleted RNA-seq library preparation kit.
- High-throughput sequencer (Illumina NovaSeq 6000 or equivalent).
Methodology:
- Cell Line Engineering: Create three stable cell lines using CRISPR/Cas9: (i) Wild-type control, (ii) p150-specific knockout (retaining p110 expression), (iii) Complete ADAR1 knockout.
- Treatment: Treat wild-type and p150-KO cells with Poly(I:C) (1 µg/mL, 24h) to induce interferon response and p150 expression.
- RNA Extraction & Sequencing: Harvest cells. Isolate total RNA, ensure high RIN (>8.5). Prepare strand-specific, rRNA-depleted libraries. Sequence to a minimum depth of 50 million paired-end 150bp reads per sample.
- Computational Analysis: a. Alignment: Trim reads (Trim Galore!) and align to the human reference genome (GRCh38) using a splice-aware aligner (STAR). b. Editing Detection: Process BAM files using REDItools2 or JACUSA2 in "difference" mode between RNA-seq data and the reference genome. Apply strict filters: remove known SNPs (dbSNP), require minimum read depth (≥10), and editing frequency (≥0.1). c. Isoform Attribution: Classify filtered editing sites: * p150-dependent: Sites where editing is lost in p150-KO cells after Poly(I:C) treatment but remains in similarly treated wild-type cells. * p110-dependent (basal): Sites where editing is significantly reduced in complete ADAR1 KO versus wild-type, even without Poly(I:C) treatment. * Shared/Cooperative: Sites that show reduced but not absent editing in either single-isoform perturbation, suggesting functional redundancy.
Diagram 1: Workflow for Isoform-Specific ADAR1 Editing Analysis.
Signaling Pathways in ADAR1 Isoform Regulation
The differential activity of ADAR1 isoforms is governed by distinct upstream signals.
Diagram 2: Signaling Pathways Driving ADAR1 p150 vs p110 Expression.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for ADAR1 Isoform-Specific Research
| Reagent / Material | Function in Experiment | Key Consideration |
|---|---|---|
| Isoform-Specific CRISPR Guides | To genetically ablate only the p150 isoform (via its promoter) or the p110 isoform (via its unique first exon). | Requires careful design and validation by qPCR (isoform-specific primers) and western blot. |
| Poly(I:C) High Molecular Weight | A synthetic dsRNA analog used to robustly induce interferon response and upregulate ADAR1 p150 expression. | Concentration and transfection method (e.g., lipofection vs. electroporation) must be optimized per cell type. |
| Strand-Specific, rRNA-depletion Kit (e.g., Illumina Stranded Total RNA Prep) | For RNA-seq library prep that removes abundant rRNA and preserves strand information, crucial for detecting editing in antisense transcripts. | Superior to poly-A selection for capturing non-coding RNAs and incompletely spliced transcripts, common editing targets. |
| ADAR1 p150 & p110 Specific Antibodies | For western blot validation of isoform expression changes post-perturbation or treatment. | Many commercial antibodies detect both isoforms; seek ones validated for isoform-specificity (e.g., targeting unique N-termini). |
| Validated siRNA for Total ADAR1 | A positive control for complete loss of A-to-I editing in functional assays. | Ensures observed phenotypes are ADAR1-dependent. |
RStudio with Bioconductor Packages (e.g., Reditools, RISA) |
For downstream statistical analysis, visualization, and differential editing detection. | Enables reproducible analysis and integration with other omics datasets (e.g., differential gene expression). |
Navigating Experimental Pitfalls: Challenges in Distinguishing p110 from p150 Function
Cross-Reactivity and Validation of Isoform-Specific Reagents
This guide is published within the context of a broader research thesis comparing the functional roles of the ADAR1 p110 (constitutive, interferon-independent) and p150 (inducible, interferon-dependent) isoforms in RNA editing, innate immune response modulation, and implications in cancer and autoimmunity. The accurate, isoform-specific detection and manipulation of these proteins are critical for elucidating their distinct functions. This comparison guide objectively evaluates the performance of key commercially available isoform-specific reagents against common alternatives, supported by experimental validation data.
Comparative Analysis of ADAR1 Isoform-Specific Antibodies
The table below summarizes validation data for leading anti-ADAR1 antibodies, focusing on their specificity for p110 versus p150 isoforms as determined by siRNA knockdown and overexpression experiments.
Table 1: Performance Comparison of ADAR1 Isoform-Specific Antibodies
| Vendor & Catalog # | Target Epitope / Clone | Claimed Specificity | Experimental Validation (Knockdown/WB) | Cross-Reactivity Observed? | Key Application (Validated) |
|---|---|---|---|---|---|
| Vendor A, #AB1234 | N-terminus, p150-specific | p150 only | siRNA to p150 ablates signal; p110 KO cells show signal. | No cross-reactivity with p110. | Western Blot, IF |
| Vendor B, #SC5678 | C-terminus, common region | p110 & p150 | Detects both isoforms; signal lost with total ADAR1 KO. | Not applicable (pan-specific). | IP, Western Blot |
| Vendor C, #CD9012 | Internal, p110-specific | p110 only | p110 siRNA eliminates signal; p150 induction does not alter signal. | Minimal (≤5% cross-reactivity with p150). | Western Blot |
| Vendor D, #EF3456 | Z-DNA binding domain | p150 preferential | p150 knockdown reduces but does not abolish signal. | Yes (~30% residual signal from p110). | Western Blot |
Supporting Experimental Protocol: Western Blot Validation of Specificity
- Cell Lines: Wild-type, ADAR1 p110 knockout (genetically engineered), and ADAR1 total knockout HEK293T cells.
- Treatment: Cells were treated with 500 U/mL interferon-α (IFN-α) for 24 hours to induce p150 expression.
- Lysis & Protein Quantification: Cells were lysed in RIPA buffer with protease inhibitors. Protein concentration was determined via BCA assay, and 30 µg of total protein was loaded per lane.
- Gel Electrophoresis: Proteins were separated on a 4-12% Bis-Tris polyacrylamide gel.
- Transfer & Blocking: Transferred to PVDF membrane, blocked with 5% non-fat milk in TBST.
- Antibody Incubation: Membranes were probed with target antibodies (Table 1) at 1:1000 dilution overnight at 4°C, followed by HRP-conjugated secondary antibody (1:5000) for 1 hour.
- Detection: Chemiluminescent substrate was applied, and signals were captured. Membranes were subsequently stripped and re-probed with a β-actin loading control antibody.
- Key Control: Lysates from p110 KO cells + IFN-α treatment provide a clean background to assess p150-specific antibody performance.
Comparison of CRISPR/Cas9 Strategies for Isoform-Specific Knockout
Genetic tools are essential for functional studies. The table compares two common strategies for generating isoform-specific ADAR1 knockouts.
Table 2: Comparison of Genetic Editing Strategies for ADAR1 Isoforms
| Strategy | Target Genomic Region | Expected Outcome | Validation Method | Efficiency & Notes |
|---|---|---|---|---|
| Exon Skipping (p110-specific) | Intron-exon boundary of exon 2 (exclusive to p110 transcript). | Frameshift mutation or exon skipping, ablating p110 protein synthesis. p150 remains intact. | Western Blot with p110-specific antibody. RT-PCR of edited region. | High specificity (>95%). Requires careful sgRNA design to avoid disrupting p150-specific promoter/exon 1A. |
| Promoter/Exon 1A Targeting (p150-specific) | Unique first exon (Exon 1A) or promoter of the interferon-inducible transcript. | Disruption of p150 transcription initiation. p110 expression unaffected. | Western Blot pre- and post-IFN-α induction using p150-specific antibody. qRT-PCR for p150 transcript. | Specific, but efficiency depends on chromatin state. Must confirm no impact on proximal p110 promoter. |
Supporting Experimental Protocol: Validation of p150-Specific Knockout via qRT-PCR
- Cell Transfection & Selection: HEK293T cells were transfected with a CRISPR/Cas9 plasmid containing a guide RNA targeting the p150-specific exon 1A. Puromycin selection was applied for 72 hours.
- IFN-α Induction: Pooled selected cells were treated with 500 U/mL IFN-α for 24 hours to induce p150 expression.
- RNA Extraction & cDNA Synthesis: Total RNA was extracted using a column-based kit. 1 µg of RNA was reverse transcribed using random hexamers.
- qPCR Primers: Two primer sets were used:
- Set 1 (p150-specific): Forward primer in exon 1A, reverse in constitutive exon.
- Set 2 (p110-specific): Forward primer in constitutive exon 1B, reverse in constitutive exon.
- Set 3 (Control): GAPDH housekeeping gene.
- qPCR Reaction: SYBR Green Master Mix was used. Cycling conditions: 95°C for 10 min, followed by 40 cycles of 95°C for 15 sec and 60°C for 1 min.
- Analysis: The ΔΔCt method was used to quantify relative expression of p150 and p110 transcripts in edited cells versus wild-type IFN-α-treated controls.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for ADAR1 Isoform Research
| Item | Function & Relevance | Example Product/Catalog |
|---|---|---|
| p110-Specific Antibody | Detects the constitutive 110 kDa isoform in Western Blot/IF without cross-reacting with p150. Critical for assays without interferon stimulation. | Vendor C, #CD9012 |
| p150-Specific Antibody | Detects the inducible 150 kDa isoform. Must be validated for use in interferon-stimulated conditions. | Vendor A, #AB1234 |
| Pan-ADAR1 Antibody | Recognizes a common epitope in both isoforms. Useful for quantifying total ADAR1 protein levels. | Vendor B, #SC5678 |
| Recombinant Human IFN-α | Induces expression of the p150 isoform via the JAK-STAT pathway. Essential for p150 functional studies. | PeproTech, #300-02A |
| ADAR1 p110 Knockout Cell Line | Ready-made model to study p150 function in isolation or to validate p150-specific reagents. | Horizon Discovery, HZGHC000864c011 |
| p150-Specific CRISPR/Cas9 Kit | Pre-designed sgRNA and Cas9 components for generating a p150-specific knockout cell line. | Santa Cruz Biotechnology, sc-400659 |
| Inosine-Specific RNA Sequencing Service | Definitive method to map RNA editing sites globally and distinguish editing activity attributable to each isoform. | Next-Gen Sequencing provider with inosine detection pipeline. |
Visualization of Key Methodologies and Pathways
ADAR1 Isoform-Specific Antibody Validation Workflow
IFN-α Induction of ADAR1 p150 Signaling Pathway
CRISPR Strategy for p110 vs. p150 Knockout
Interpreting experimental data in ADAR1 research requires careful consideration of compensatory mechanisms activated when one isoform (p110 or p150) is lost. This guide compares methodologies and data interpretation for studying isoform-specific functions, framed within the broader thesis of ADAR1 p110 versus p150 functional comparison.
Key Experimental Comparisons & Data
The following table summarizes core experimental approaches and typical quantitative outcomes when investigating compensation between ADAR1 p110 and p150 isoforms.
Table 1: Experimental Approaches & Outcomes in ADAR1 Isoform-Specific Studies
| Experimental Manipulation | Primary Measured Outcome | Key Findings (Typical Data Range) | Interpretation Challenge |
|---|---|---|---|
| p150-specific knockout/knockdown | Global A-to-I editing levels (e.g., in Alu elements) | Editing reduction: 60-80% in immune-activated cells. Editing in specific non-Alu sites may be preserved (<20% reduction). | p110 may partially compensate at a subset of structured RNA sites. |
| p110-specific knockout/knockdown | Global A-to-I editing levels | Editing reduction: 10-30% in most cell types. Significant reduction in specific housekeeping transcripts. | p150 (constitutively expressed) may maintain bulk editing; cell type-specific effects are critical. |
| Double p110/p150 knockout | Cell viability, interferon response, editing | Near-total editing loss (>95%). Lethal in vivo. Strong constitutive interferon activation. | Establishes the non-redundant essential function of total ADAR1. |
| p150-only expression (p110 KO) | Interferon pathway activation (e.g., ISG mRNA) | ISG upregulation: 5-50 fold increase vs. wild-type, depending on cell state. | Reveals p110's unique role in preventing MDA5 sensing of endogenous dsRNA in cytoplasm. |
| p110-only expression (p150 KO) | Response to immune activation (e.g., poly I:C, IFN) | Editing of inducible sites impaired. Enhanced cell death upon viral mimic treatment. | Highlights p150's essential, inducible role in immune signaling contexts. |
| Editing site-specific RNA-seq | Site-specific editing ratios (A-to-I %) | Divergent patterns: Some sites edited exclusively by one isoform, others show shared responsibility with ratio shifts upon KO. | Distinguishing direct loss from indirect compensatory regulation requires controlled kinetics. |
Detailed Experimental Protocols
Protocol 1: Isoform-Specific Knockout & RNA Editing Analysis Objective: To assess editing changes and compensatory upregulation upon loss of one ADAR1 isoform.
- Cell Line Engineering: Use CRISPR-Cas9 to generate isogenic cell lines with frameshift mutations in exon-specific regions for ADAR1 p110 (exon 2 targeting) or p150 (exon 1 targeting). Validate by western blot (p110: ~110 kDa; p150: ~150 kDa) and RT-qPCR with isoform-specific primers.
- RNA Sequencing: Isolate total RNA (triplicate biological replicates) from wild-type and knockout lines under basal and interferon-beta (1000 U/mL, 24h) treated conditions. Perform 150bp paired-end sequencing on poly-A selected libraries.
- Editing Analysis: Align RNA-seq reads to the reference genome (STAR aligner). Use specialized variant callers (e.g., REDItools2, JACUSA2) to identify A-to-G/T-to-C mismatches. Filter against common SNPs (dbSNP). Quantify editing levels (percentage) at known ADAR targets (e.g., Alu elements, curated non-repetitive sites).
- Compensation Assessment: Compare global editing levels. Perform differential editing analysis. Quantify mRNA expression of the remaining ADAR1 isoform via RNA-seq counts to check for transcriptional upregulation.
Protocol 2: Cytoplasmic dsRNA Detection & Interferon Response Objective: To measure the functional consequence of isoform loss and identify compensatory suppression of interferon signaling.
- Immunofluorescence for dsRNA: Culture isoform-specific KO cells on chamber slides. Fix with 4% paraformaldehyde, permeabilize with 0.1% Triton X-100. Stain with J2 anti-dsRNA antibody (1:500) overnight at 4°C, followed by fluorescent secondary antibody. Counterstain nuclei with DAPI. Image using confocal microscopy.
- Interferon-Stimulated Gene (ISG) Quantification: Treat cells (wild-type, p110-KO, p150-KO) with poly(I:C) (1 µg/mL, transfected via lipofectamine) for 6h. Extract RNA and synthesize cDNA. Perform RT-qPCR for canonical ISGs (e.g., IFIT1, ISG15, MX1) using SYBR Green assays. Normalize to GAPDH or ACTB. Calculate fold change vs. untreated wild-type using the 2^(-ΔΔCt) method.
- Data Interpretation: Elevated cytoplasmic dsRNA signal and concomitant ISG induction in p110-KO cells indicate failed compensation by p150 for this function. Minimal ISG activation in p150-KO under basal conditions suggests effective p110 compensation for preventing MDA5 activation.
Visualizing ADAR1 Isoform Regulation & Compensation
Title: ADAR1 Isoform Roles in dsRNA Editing & Immune Prevention
Title: Experimental Knockout Outcomes Summary
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for ADAR1 Isoform-Specific Research
| Reagent/Material | Function & Application | Key Consideration |
|---|---|---|
| Isoform-Specific Antibodies | Western blot, Immunofluorescence. Distinguish p150 (N-terminal Z-DNA binding domain) from p110. | Validate specificity using isoform-KO cell lysates. Commercial p150-specific antibodies are limited. |
| CRISPR gRNAs (Exon-Specific) | Generation of isoform-specific knockout cell lines. Target exon 1 for p150, exon 2 or downstream for p110. | Requires careful off-target prediction and sequencing validation of edits. |
| J2 anti-dsRNA Antibody | Immunofluorescence & dot blot to detect cytoplasmic dsRNA accumulation, a marker of editing deficiency. | Recognizes dsRNA >40bp; confirms functional loss of editing activity. |
| Poly(I:C) (HMW) | A synthetic dsRNA viral mimic. Transfect to induce innate immune response and test p150's inducible role. | Use with appropriate transfection reagent for cytoplasmic delivery (e.g., Lipofectamine 2000). |
| Interferon-beta | Treatment to induce p150 expression via the JAK-STAT pathway. Used to study regulated vs. constitutive editing. | Typical research concentration range: 500-1000 U/mL for 12-24h. |
| RNA-seq Library Prep Kits (Ribo-Depletion) | For comprehensive editing analysis, including non-polyadenylated Alu transcripts. Poly-A selection will miss key substrates. | Use kits like Illumina's Ribo-Zero Plus to retain non-coding RNA. |
| Editing-Specific Bioinformatics Pipelines | Tools like REDItools2, JACUSA2, or SPRINT to identify A-to-I editing sites from RNA-seq data. | Must be paired with matched genomic DNA sequencing or stringent filters to exclude SNPs. |
| Isoform-Specific RT-qPCR Primers | Quantify transcript levels of individual isoforms to assess compensatory transcriptional upregulation. | Design primers spanning unique exon junctions (e.g., p150-specific: exon 1A to exon 2). |
Within the critical context of ADAR1 p110 versus p150 isoform functional research, a core methodological challenge is the misinterpretation of data due to overexpression artifacts. This guide compares experimental approaches for the accurate subcellular localization and RNA editing analysis of these isoforms, highlighting how overexpression can generate misleading signals and providing protocols for robust comparison.
Comparative Analysis of Experimental Approaches
Table 1: Comparison of ADAR1 Isoform Expression & Localization Methods
| Method | Key Principle | Fidelity for p110/p150 Localization | Risk of Artifact | Typical Throughput | Key Quantitative Output |
|---|---|---|---|---|---|
| Transient Plasmid Overexpression | High-level expression from CMV/SV40 promoters. | Low - Saturation masks native regulation. | Very High - Cytoplasmic aggregation; nuclear mislocalization. | High | Fluorescence intensity (arbitrary units). |
| Stable Genomic Integration (Baculoviral System) | Single-copy integration at a defined locus. | Moderate-High - Physiological expression levels. | Low | Medium | Molecules/cell; precise localization ratio. |
| Endogenous Tagging (CRISPR-Cas9) | Tagging the native genomic locus. | Very High - Preserves all regulatory elements. | Very Low | Low (per cell line) | Native abundance and localization. |
| In Situ Hybridization (RNA) | Detection of endogenous transcript. | High for RNA localization. | Low (if probes are specific) | Medium | Transcript count per compartment. |
Table 2: Impact of Expression Method on Observed Editing Signals
| Expression Method | Observed Global Editing Hyperediting (Promiscuous Editing) | Site-Specific Editing Fidelity | Key Artifact Source |
|---|---|---|---|
| High-Level p150 Overexpression | Dramatically increased | Reduced specificity; off-targets increase. | Saturation of endogenous dsRNA; disruption of p110:p150 balance. |
| High-Level p110 Overexpression | May increase or decrease artificially. | May be unrepresentative of nuclear activity. | Mislocalization to cytoplasm; competition with endogenous p150. |
| Physiological Expression (Stable/Endogenous) | Represents native activity. | High - Reflects true isoform-specific roles. | Minimal. |
Experimental Protocols for Artifact-Free Comparison
Protocol 1: Quantitative Subcellular Localization with Controlled Expression
Objective: To accurately determine the nuclear-to-cytoplasmic (N:C) ratio of ADAR1 p110 and p150 isoforms while avoiding saturation artifacts.
- Construct Design: Clone ADAR1 p110 and p150 cDNAs into a vector system permitting stable, single-copy genomic integration (e.g., BAC transgenesis or Flp-In T-REx). Use a C-terminal tag (e.g., HALO, mEGFP).
- Cell Line Generation: Generate isogenic cell lines with integrated, inducible constructs. Use a low-strength, inducible promoter (e.g., tetracycline-responsive) to enable titration.
- Induction & Titration: Induce expression with a titrated dose of doxycycline (e.g., 0-100 ng/mL) for 24 hours. Aim for expression levels within 2x of endogenous (validate by Western).
- Imaging & Quantification:
- Fix cells and stain nucleus (DAPI).
- Acquire confocal images under non-saturating conditions.
- Use image analysis software (e.g., CellProfiler) to define nuclear and cytoplasmic masks.
- Calculate the mean fluorescence intensity in each compartment. Report the N:C ratio for at least 200 cells per condition.
- Controls: Include a line with endogenously tagged ADAR1 (CRISPR-Cas9) as the gold-standard control.
Protocol 2: Editing-Specific RNA Sequencing (Edit-seq) from Physiological Expression
Objective: To profile isoform-specific RNA editing events without hyperediting artifacts.
- Sample Preparation: Use cell lines from Protocol 1 induced to near-physiological levels. Include a negative control (ADAR1 knockout line).
- RNA Isolation & Library Prep: Isolve total RNA. Perform poly-A selection. Prepare stranded RNA-seq libraries. Crucially, do not use rRNA depletion, as it can enrich for hyperedited transcripts that do not polyadenylate.
- Sequencing: Perform deep sequencing (≥50 million paired-end 150bp reads per sample).
- Bioinformatic Analysis:
- Map reads to the genome using a splice-aware aligner (STAR) in two-pass mode.
- Identify editing sites using dedicated tools (e.g., REDItools2, JACUSA2) with stringent filters.
- Filter sites against common SNPs (dbSNP).
- Key Metric: Calculate the Editing Index (number of confirmed A-to-I edits per million aligned reads in Alu/non-Alu regions) for each isoform-expressing line versus knockout.
- Validation: Validate top candidate sites using amplicon sequencing (Sanger or Illumina).
Visualization of Pathways and Workflows
Title: Overexpression Artifacts vs. Physiological Expression Workflow
Title: ADAR1 p110 and p150 Isoform Generation and Function
Title: Edit-seq Experimental Workflow for Artifact-Free Data
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Artifact-Minimized ADAR1 Research
| Item | Function in p110/p150 Research | Key Consideration for Avoiding Artifacts |
|---|---|---|
| Inducible Expression System (e.g., Flp-In T-REx) | Enables single-copy, titratable expression of tagged isoforms. | Prevents overexpression; allows dose-response to find physiological levels. |
| HALO or SNAP-tag Ligands | For highly specific, covalent labeling of tagged proteins for imaging. | Reduces background vs. traditional fluorescent proteins; enables pulse-chase. |
| Baculoviral Gene Delivery System | For efficient, single-copy delivery of large constructs to diverse cells. | Achieves consistent, stable expression without random multi-copy integration. |
| CRISPR-Cas9 Knock-in Reagents | For endogenously tagging the ADAR1 locus. | The gold standard for preserving all native expression regulation. |
| Stranded Poly-A Selection Kit | For RNA-seq library preparation focused on mature mRNAs. | Avoids enrichment of hyperedited nuclear RNAs that lack poly-A tails. |
| Specific Antibodies (p110 vs p150) | For immunoblotting and immunofluorescence of endogenous proteins. | Validates expression levels; must be isoform-specific (target unique N-terminus). |
| dsRNA Sensor Cell Line (e.g., MDA5-knock-in reporter) | Functional assay for cytoplasmic dsRNA accumulation. | Measures functional consequence of p150 activity/knockout without editing bias. |
Disentangling Cell-Autonomous from Immune-Mediated Phenotypes In Vivo
Publish Comparison Guide: ADAR1 Isoform-Specific Mouse Models forIn VivoPhenotyping
Understanding whether a phenotype is intrinsically driven by a gene defect within a cell (cell-autonomous) or results from extrinsic immune pressure is a fundamental challenge. This guide compares the performance of mouse models specifically engineered to disrupt ADAR1 isoforms p110 or p150 in this experimental context.
Core Model Comparison & Phenotypic Data
Table 1: Comparative *In Vivo Outcomes of ADAR1 Isoform-Specific Disruption*
| Model / Genetic Alteration | Key Immune-Mediated Phenotype | Key Cell-Autonomous Phenotype | Lethality | Rescue by Immune Deficiency | Supporting Experimental Data |
|---|---|---|---|---|---|
| Global p150-only knockout(Disruption of cytoplasmic isoform) | Severe multi-organ inflammation; T-cell & macrophage infiltration. | Embryonic lethality ~E12.5. | Complete (E12.5). | Yes. Crossing to Rag1-/- or Ifnar1-/- rescues embryonic lethality and permits live birth. | RNA-seq & Ribo-seq data show MDA5 activation and translational shutdown in double mutants (Adar1-/- Mavs-/-) but not in Adar1-/- Mavs+/+, isolating cell-autonomous effects. |
| Global p110-only knockout(Disruption of nuclear isoform) | Mild or absent inflammation. | Perinatal lethality; defective erythropoiesis. | Perinatal (P0-P1). | No. Lethality persists in Rag1-/- background. | Hematopoietic stem cell (HSC) transplant studies show non-cell-autonomous support requirement; fetal liver analysis reveals intrinsic erythroid defects. |
| Conditional, tissue-specific p150 deletion(e.g., in intestinal epithelium) | Severe enteritis; immune cell recruitment. | Epithelial integrity loss, apoptosis. | Context-dependent. | Partial. Immune deficiency reduces tissue inflammation but intrinsic epithelial defects persist. | Parabiosis or bone marrow chimera experiments show phenotype is primarily tissue-intrinsic, exacerbated by immune recruitment. |
| A-to-I Editing-Deficient Point Mutant (E861A)(Disables editing for both isoforms) | Lethal interferonopathy, massive ISG induction. | Embryonic lethality ~E13.5. | Complete (E13.5). | Yes. Rescued by Mavs-/-. | Directly proves that loss of RNA editing, not protein interaction loss, drives immune activation. |
Detailed Experimental Protocols
1. Protocol for Disentangling Phenotypes via Immune-Deficient Crosses
- Objective: To determine if a lethal or pathological phenotype is caused by immune system attack.
- Method:
- Cross the ADAR1 isoform-specific mutant mouse (e.g., Adar1p150-/-) with a globally immune-deficient strain (e.g., Rag1-/- [lacks T/B cells] or Ifnar1-/- [insensitive to Type I IFN]).
- Generate double-mutant offspring.
- Compare viability, histopathology, and molecular readouts (e.g., ISG expression via qPCR) between single mutants and double mutants.
- Interpretation: Rescue of lethality and inflammation in the double mutant indicates an immune-mediated phenotype. Persistence of a phenotype indicates a cell-autonomous defect.
2. Protocol for Cell-Autonomous Analysis via In Vitro Culture & Transplantation
- Objective: Isolate intrinsic cellular function of ADAR1 isoforms devoid of in vivo immune cues.
- Method:
- Isolate primary cells (e.g., HSCs, fibroblasts) from E12.5-E14.5 Adar1p110-/- or Adar1p150-/- embryos.
- Perform competitive proliferation/apoptosis assays in vitro.
- For hematopoietic lineages, perform bone marrow transplantation of mutant cells into lethally irradiated, immunocompetent wild-type hosts. Alternatively, transplant wild-type bone marrow into irradiated mutant hosts.
- Interpretation: Defects in mutant cells in culture or when competing in a wild-type host are cell-autonomous. Defects only seen in mutant hosts receiving wild-type cells indicate a non-cell-autonomous, microenvironmental defect.
Mandatory Visualizations
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for ADAR1 In Vivo Phenotyping Studies
| Research Reagent | Function & Application | Example/Supplier |
|---|---|---|
| Isoform-Specific Knockout Mice | In vivo models to dissect p110 vs. p150 function. Generated via CRISPR or traditional targeting. | Adar1E861A (edit-dead), Adar1p150-/-, conditional floxed alleles. |
| Immodeficient Mouse Strains | Genetic backgrounds to suppress immune response and isolate cell-autonomous effects. | Rag1-/-, Ifnar1-/-, Mavs-/-, Stat1-/-. |
| Anti-ADAR1 Antibodies (Isoform-specific) | Detect and localize p110 and p150 by WB, IHC, IF. Critical for validating models. | Commercial clones: sc-73408 (Santa Cruz), ab126745 (Abcam) - require validation for isoform specificity. |
| Anti-dsRNA Antibody (J2) | Detect unedited immunogenic dsRNA substrates that accumulate upon ADAR1 loss. | J2 monoclonal antibody (SCICONS). Standard for immunofluorescence and dot blot. |
| Type I IFN Reporter Mice | Visualize and quantify in vivo interferon response activation spatially and temporally. | Mx1-GFP, ISG-GFP (e.g., ISG15-Luc) reporter strains. |
| Next-Gen Sequencing Kits | Profile transcriptome-wide (RNA-seq) and editome (TREAT, SAILOR) changes. | Illumina TruSeq, protocols for editing-sensitive RNA-seq. |
| MDA5/RLR Pathway Inhibitors | Pharmacological tools to validate mechanism in vitro or ex vivo. | Ruxolitinib (JAK inhibitor), CVA (MDA5 inhibitor). |
Optimizing Conditions to Distinguish Basal (p110) from Induced (p150) Editing Activity
Within ADAR1 isoform research, a central challenge is the precise differentiation of editing activity driven by the constitutively expressed, interferon-independent p110 isoform from that of the interferon-induced p150 isoform. This distinction is critical for understanding innate immune regulation, viral response, and dysregulation in diseases like cancer and autoimmunity. This guide compares experimental strategies and their efficacy in isolating and quantifying isoform-specific adenosine-to-inosine (A-to-I) editing.
Comparative Analysis of Methodological Approaches
Table 1: Comparison of Core Methodological Strategies
| Method / Condition | Core Principle | p110 Specificity | p150 Specificity | Key Experimental Readout | Major Limitation |
|---|---|---|---|---|---|
| Interferon Stimulation | Induces p150 expression; compare editing pre/post IFN. | Low (measures change) | High | Editing rate increase at selective sites (e.g., 3' UTR Alu elements). | Global cellular changes; p110 levels may also be modulated. |
| p110-Specific Knockout/Conditional KO | Genetic removal of p110 while retaining p150 expression potential. | High (defines p110-ablated baseline) | Indirect | Residual editing in KO after IFN = p150 activity. | Requires complex cell/animal models; compensatory mechanisms. |
| Cytoplasmic vs. Nuclear Fractionation | Exploits p150's cytoplasmic localization vs. p110's nuclear. | Moderate (nuclear fraction) | Moderate (cytoplasmic fraction) | Subcellular editing profiles. | Cross-contamination risk; editing sites can be in both compartments. |
| In vitro Editing Assay with Recombinant Proteins | Purified p110 vs. p150 proteins assayed on defined substrates. | Direct | Direct | Kinetic parameters (Km, kcat) on specific RNA structures. | May not reflect cellular context, localization, or co-factor interactions. |
| Selective Inhibitor Profiling | Use of compounds that show isoform selectivity. | Dependent on inhibitor | Dependent on inhibitor | Differential inhibition curves (IC50). | Few highly selective tool compounds available. |
Table 2: Quantitative Data from Representative Studies
| Study Model | Condition/Intervention | p110-Associated Editing Signal | p150-Associated Editing Signal | Key Metric (Fold Change/ΔEditing %) |
|---|---|---|---|---|
| WT vs. ADAR1 p150-only (Cell line) | IFN-β treatment (24h) | Minimal change | +8.2 fold | Increase in editing at AZIN1 3'UTR site (from 5% to 41%). |
| Cytoplasmic Fractionation (HEK293T) | IFN-γ treatment | Nuclear editing: +15% | Cytoplasmic editing: +320% | Alu element editing in cytoplasmic RNA. |
| Recombinant Protein Assay | In vitro on dsRNA | kcat = 2.1 min⁻¹ | kcat = 5.8 min⁻¹ | Catalytic efficiency (kcat/Km) for p150 ~2.7x higher on long dsRNA. |
| p110-KO MEFs | Basal vs. IFN-α | 98% of basal editing lost | Rescue of 30% of sites post-IFN | Site-selective, IFN-induced editing recovery. |
Detailed Experimental Protocols
Protocol 1: Distinguishing Activity via Interferon Induction and RNA-Seq
Objective: To identify p150-induced editing events by transcriptional induction.
- Cell Culture & Treatment: Seed appropriate cells (e.g., A549, HeLa) in triplicate. Treat one set with human IFN-β (1000 U/mL) for 24-48 hours. Maintain a parallel untreated set.
- RNA Extraction & Quality Control: Harvest cells using TRIzol. Isolve total RNA. Assess integrity via RIN > 8.5 (Bioanalyzer).
- Library Preparation & Sequencing: Deplete ribosomal RNA. Prepare stranded RNA-seq libraries. Sequence on an Illumina platform to a depth of ≥50 million paired-end 150bp reads per sample.
- Bioinformatic Analysis:
- Map reads to the human reference genome (e.g., GRCh38) using splice-aware aligners (STAR).
- Identify A-to-I editing sites using specialized callers (e.g., REDItools2, JACUSA2) with stringent filters (e.g., ≥10 reads coverage, editing level ≥1%, mismatch frequency >> sequencing error rate).
- Key Comparison: Calculate the differential editing rate (DER) between IFN-treated and untreated samples. Genuine p150-induced sites will show a significant increase in editing level (e.g., Fisher's exact test, FDR < 0.05) and are often enriched in Alu repeats within 3' UTRs.
- Validation: Validate top candidate sites via amplicon sequencing (PCR on cDNA, then deep sequencing).
Protocol 2:In VitroEditing Assay with Purified Isoforms
Objective: To directly compare the enzymatic activity of purified p110 and p150 on defined RNA substrates.
- Protein Purification: Express and purify recombinant human ADAR1 p110 and p150 (containing catalytic domain) with N-terminal tags (e.g., GST, 6xHis) from insect or mammalian expression systems.
- RNA Substrate Preparation: Synthesize short (e.g., 50-100 bp) dsRNA substrates by in vitro transcription. Incorporate a single target adenosine at a known position. Radiolabel (α-32P-ATP) or fluorophore-label the strand.
- Kinetic Reaction Setup: Set up reactions containing reaction buffer (e.g., 25 mM HEPES-KOH pH 7.0, 100 mM KCl, 5% glycerol, 1 mM DTT, 0.1 mg/mL BSA), labeled substrate (concentration series from 10 nM to 1 μM), and a fixed, low concentration of purified enzyme (e.g., 10 nM).
- Reaction & Quenching: Incubate at 30°C. Remove aliquots at time points (e.g., 0, 1, 2, 5, 10, 20 min) and quench with 3x volume of stop solution (90% formamide, 50 mM EDTA).
- Product Analysis: Resolve reaction products by denaturing urea-PAGE. Quantify the proportion of edited (cleaved by Endonuclease V or mobility shifted) vs. unedited substrate using a phosphorimager.
- Data Analysis: Plot initial velocity vs. substrate concentration. Fit data to the Michaelis-Menten equation to determine kinetic parameters (Km, Vmax, kcat).
Visualizations
Diagram 1: ADAR1 p110 vs p150 Domain Structure and Localization
Diagram 2: Experimental Workflow for IFN-based Distinction
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Distinguishing p110/p150 Activity | Key Considerations |
|---|---|---|
| Recombinant Human IFN-β/γ | Gold-standard inducer of p150 expression. | Use validated biological activity units; optimize dose (100-1000 U/mL) and time (18-48h) for your cell type. |
| ADAR1 Isoform-Specific Antibodies | Detect and quantify protein levels via WB, IF. | Confirm specificity for p110 (N-terminal) vs. p150 (Zα domain). Critical for validation. |
| p110-floxed / Conditional KO Models | Genetically isolate p150 function by ablating constitutive p110. | Enables clean baseline establishment. Available as cell lines (e.g., MEFs) or mice. |
| Cytoplasmic & Nuclear RNA Kits (e.g., PARIS Kit) | Physically separate RNA substrates by compartment. | Essential for localization-based assays. Validate fraction purity with compartment-specific markers (e.g., MALAT1 nuclear, GAPDH cytoplasmic). |
| Recombinant p110 & p150 Proteins (Active) | Direct in vitro kinetic and substrate specificity studies. | Source from mammalian (more native) or insect (higher yield) systems. Confirm deaminase activity. |
| Selective ADAR1 Inhibitors (e.g., 8-azaadenosine derivatives) | Pharmacological perturbation to profile isoform sensitivity. | Current compounds often inhibit both; used to probe editing-dependent phenotypes. |
| Alu-rich or Structured RNA Reporters (Plasmid or synthetic) | Defined substrates for transfection-based or in vitro editing assays. | Design based on known endogenous targets (e.g., AZIN1 3'UTR). Allows precise quantification. |
p110 vs. p150: A Direct Functional Comparison in Editing, Immunity, and Disease
Within the broader investigation of ADAR1 p110 versus p150 isoform functions, a critical question pertains to their inherent catalytic activity and substrate preference. This guide compares the in vitro editing catalysis performance of recombinant ADAR1 p110 and p150 isoforms on defined RNA substrates, synthesizing current experimental data to delineate overlap and specificity.
Comparative Catalytic Performance Data
The following table summarizes key quantitative findings from recent in vitro editing assays comparing purified human ADAR1 isoforms. Data is normalized to the p150 activity on a perfect duplex dsRNA substrate.
Table 1: In Vitro Editing Catalysis Comparison of ADAR1 Isoforms
| Parameter | ADAR1 p110 | ADAR1 p150 | Notes / Experimental Condition |
|---|---|---|---|
| Activity on Perfect Duplex dsRNA | 30-40% | 100% (Reference) | Long (>50 bp) perfectly base-paired RNA; measured by conversion rate. |
| Activity on Short Stem-Loop (e.g., miR-376) | 10-15% | 80-90% | Structured endogenous miRNA-type substrate. |
| Deamination Rate (kcat) on dsRNA | 1.2 ± 0.3 min⁻¹ | 3.8 ± 0.5 min⁻¹ | Single-turnover kinetics, 37°C. |
| Apparent KM for dsRNA | 180 ± 20 nM | 50 ± 10 nM | Reflects substrate binding affinity. |
| A-to-I Editing Specificity | High | Moderate | p150 shows more promiscuous editing at non-canonical sites in long dsRNA. |
| Z-DNA/RNA Binding Domain Effect | Not Applicable | +30-50% Activity | p150 activity is enhanced on substrates co-presenting Z-form nucleic acid. |
| Response to 2'-O-methylation | Inhibited (>70% loss) | Partially Inhibited (~40% loss) | Substrate modification mimicking viral defense. |
Detailed Experimental Protocols
Protocol 1: Recombinant Protein Purification for Catalytic Assays
Objective: Produce catalytically active, purified ADAR1 isoforms.
- Express N-terminally tagged (e.g., His₆-SUMO) human ADAR1 p110 (amino acids 1-743) and p150 (amino acids 1-1226) in S. cerevisiae BJ5464 or insect cells.
- Lyse cells in buffer A (50 mM HEPES pH 7.5, 500 mM NaCl, 10% glycerol, 1 mM TCEP) with protease inhibitors.
- Perform Ni²⁺-NTA affinity chromatography. Elute with buffer A containing 250 mM imidazole.
- Cleave the tag using Ulp1 protease (for SUMO) overnight at 4°C.
- Apply to heparin-Sepharose column. Elute with a linear NaCl gradient (0.1-1.0 M) in 50 mM HEPES pH 7.5, 10% glycerol.
- Concentrate, aliquot, flash-freeze, and store at -80°C. Verify purity by SDS-PAGE and activity by standard assay.
Protocol 2: In Vitro Deamination Kinetics Assay
Objective: Measure single-turnover deamination rate constants (kobs).
- Substrate Preparation: Generate 5'-³²P-end-labeled RNA duplex (e.g., 50 bp perfect duplex from GluR-B R/G site) by annealing.
- Reaction Setup: Pre-incubate 20 nM RNA with varying enzyme concentrations (10-200 nM) in reaction buffer (20 mM HEPES pH 7.0, 100 mM KCl, 5% glycerol, 0.1 mg/mL BSA, 1 mM DTT, 0.01% NP-40) at 30°C.
- Initiation & Quenching: Start reaction by adding enzyme. Aliquots are removed at timepoints (e.g., 0, 0.5, 1, 2, 5, 10, 20 min) and quenched in 0.5% SDS/90% formamide.
- Analysis: Resolve products on denaturing 15% polyacrylamide gels. Quantify gel bands via phosphorimager. Fit the fraction unedited vs. time to a single-exponential to obtain kobs. Plot kobs vs. enzyme concentration to determine kcat and KD, RNA.
Protocol 3: High-Throughput Sequencing Specificity Analysis
Objective: Determine site-specific editing preferences across a diverse RNA library.
- Library Design: Use a synthetic RNA oligonucleotide pool containing randomized regions within a structured context.
- Editing Reaction: Incubate library with equimolar amounts of purified p110 or p150 under single-hit conditions (low enzyme:substrate ratio, short time).
- Reverse Transcription & Sequencing: Convert RNA to cDNA. Amplify with unique barcodes for each isoform reaction.
- Bioinformatics: Align sequences to reference. Calculate editing frequency (A-to-G changes) at every adenosine. Use chi-square test to identify sites with statistically significant (p<0.01) differential editing between isoforms.
Signaling & Experimental Workflow Diagrams
Diagram Title: p150-specific Z-RNA enhanced editing pathway (76 chars)
Diagram Title: Protein purification to catalytic comparison workflow (68 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for ADAR Catalysis Comparison Studies
| Item | Function in Experiment | Key Consideration |
|---|---|---|
| Recombinant ADAR1 Proteins (p110 & p150) | Core catalytic components for head-to-head assays. | Source (mammalian, insect, yeast) affects post-translational modifications and activity. Purification tags must not interfere with folding or activity. |
| Defined RNA Substrates | Validated editing targets (e.g., GluR-B R/G, miR-376, long perfect dsRNA). | Require high-purity synthesis (chemical or enzymatic) and proper annealing for duplex formation. Site-specific labeling (radioactive/fluorescent) is often needed. |
| Deamination Assay Buffer System | Provides optimal ionic and pH conditions for ADAR activity. | Typically contains HEPES/K⁺, DTT, glycerol, and carrier protein (BSA). Must be RNase-free. Chelating agents (EDTA) may be included. |
| RNase Inhibitor (e.g., RNasin) | Protects RNA substrates from degradation during incubation. | Critical for long assays or with impure protein preps. Verify compatibility with the chosen buffer system. |
| High-Throughput Sequencing Library Prep Kit | For converting edited RNA pools into sequence-ready DNA libraries. | Must minimize bias during reverse transcription and PCR amplification. Kits with unique molecular identifiers (UMIs) are preferred. |
| Z-DNA/RNA Oligonucleotides | To specifically probe p150 Zα domain function. | Chemically synthesized and HPLC-purified. Requires confirmation of left-handed Z-conformation (e.g., by CD spectroscopy). |
| Kinetic Analysis Software (e.g., Prism, KinTek Explorer) | To model single-turnover kinetics and derive kcat, KM. | Enables robust fitting of time-course data to exponential and Michaelis-Menten equations. |
This guide is framed within the ongoing research thesis comparing the ADAR1 isoforms p110 (constitutively expressed, nuclear-localized) and p150 (interferon-induced, cytoplasmic and nuclear). The central thesis investigates the distinct, non-overlapping functions of these isoforms in preventing autoinflammatory disease, with a focus on the "Gatekeeper Hypothesis." This hypothesis posits that the p150 isoform serves a critical, non-redundant role in the cytoplasm by editing endogenous double-stranded RNA (dsRNA) to prevent its recognition by the cytosolic innate immune sensor MDA5 (Melanoma Differentiation-Associated protein 5), thereby suppressing aberrant autoimmune signaling.
Comparative Performance: p110 vs. p150 in Self-RNA Sensing Suppression
The following table summarizes key experimental data comparing the functional consequences of ADAR1 p110 and p150 loss-of-function.
Table 1: Comparative Functional Analysis of ADAR1 Isoforms
| Experimental Readout | ADAR1 p110 Deficiency | ADAR1 p150 Deficiency / p150-Specific Mutation | ADAR1 Complete Knockout | Key Supporting Study |
|---|---|---|---|---|
| Subcellular Localization | Predominantly nuclear | Cytoplasmic & nuclear | N/A | Pestal et al., 2015 |
| Induction Signal | Constitutive | Type I Interferon (IFN) | N/A | George et al., 2016 |
| Basal A-to-I Editing (Cytosol) | Minimal impact | Severely reduced | Absent | Liddicoat et al., 2015 |
| MDA5 Activation (Basal) | No significant activation | Constitutive activation | Constitutive activation | Ahmad et al., 2018 |
| Interferon Signature (Basal) | Low/Normal | Highly elevated | Lethally elevated | Mannion et al., 2014 |
| Phenotype in Mice | Viable, minimal inflammation | Embryonic lethality (rescued by MDA5 or MAVS KO) | Embryonic lethality | Pestal et al., 2015; Liddicoat et al., 2015 |
| Human Disease Link | Not directly linked | Aicardi-Goutières Syndrome (AGS), Bilateral Striatal Necrosis | Dyschromatosis Symmetrica Hereditaria (DSH) | Rice et al., 2012; Herbert, 2019 |
Key Experimental Protocols
Protocol 1: Assessing MDA5 Activation via Interferon-Stimulated Gene (ISG) Expression
- Objective: Quantify the baseline activation of the MDA5 pathway in p110- vs. p150-deficient cells.
- Methodology:
- Generate isogenic cell lines (e.g., murine embryonic fibroblasts - MEFs) deficient in p110 (using targeted disruption of the interferon-inducible promoter), p150 (using knock-in of a mutation in the Z-DNA binding domain, Zα), or total ADAR1.
- Culture cells under standard conditions without exogenous interferon stimulation.
- Harvest RNA and perform quantitative RT-PCR (qRT-PCR) for canonical ISGs (e.g., Isg15, Mx1, Oas1a).
- Compare expression levels to wild-type cells. Protein levels of ISGs can be confirmed by western blot.
- Expected Data: p150-deficient cells will show a significant (~10-100 fold) increase in ISG expression compared to wild-type and p110-deficient cells, indicating constitutive MDA5 pathway activation.
Protocol 2: RNA Sequencing (RNA-seq) to Identify Unedited Alu-dsRNA Substrates
- Objective: Identify specific endogenous dsRNA substrates that are hyper-edited by p150 but not p110.
- Methodology:
- Perform total RNA extraction from cytoplasmic fractions of wild-type, p110-deficient, and p150-deficient cells.
- Prepare RNA-seq libraries. To specifically identify editing sites, use strand-specific sequencing and compare sequences to the reference genome.
- Bioinformatically pinpoint A-to-I (read as A-to-G) editing sites, focusing on regions of dsRNA formation such as inverted Alu repeats.
- Calculate the Editing Index (percentage of reads showing a G versus an A) for thousands of sites.
- Expected Data: p150-deficient cells will show a dramatic reduction in the Editing Index at specific cytoplasmic, Alu-derived dsRNA sites compared to wild-type. p110-deficient cells will show a more modest reduction, primarily at nuclear sites.
Protocol 3: Genetic Rescue of Lethality
- Objective: Test the Gatekeeper Hypothesis in vivo by determining if removing MDA5 signaling rescues the lethality of p150 deficiency.
- Methodology:
- Breed Adar1 p150-mutant mice (e.g., Adar1E861A/E861A disrupting Zα) with Mda5 knockout (Ifih1-/-) or mitochondrial antiviral-signaling protein knockout (Mavs-/-) mice.
- Genotype offspring to assess viability at weaning age.
- Analyze tissues from rescued adult mice for histological signs of inflammation.
- Expected Data: Double-mutant mice (Adar1p150-; Ifih1-/-) will be viable, demonstrating that MDA5 activation is the primary cause of embryonic lethality in p150 deficiency.
Signaling Pathway & Experimental Workflow Diagrams
Diagram 1: p150 Gatekeeper Pathway (74 chars)
Diagram 2: Genetic Rescue Experiment (68 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Investigating ADAR1 p150 Function
| Reagent / Tool | Function & Application in p150 Research | Example / Source |
|---|---|---|
| Isoform-Specific ADAR1 Antibodies | Differentiate p110 and p150 protein levels via western blot or immunofluorescence. Critical for validating knockout/depletion models. | Rabbit anti-ADAR1 p150 (Sigma-Aldrich, D7F8W); Mouse anti-ADAR1 p110 (Santa Cruz, sc-73408) |
| Zα Domain Mutant Cell Lines (E861A) | Isogenic cellular models where p150's dsRNA binding is disrupted but p110 is functional. Directly test p150's editing function. | CRISPR-engineered MEFs or human cell lines (e.g., HEK293T). |
| Cytoplasmic & Nuclear Fractionation Kits | Isolate cytoplasmic RNA to specifically analyze p150's substrate pool and prevent nuclear-editing contamination. | NE-PER Nuclear and Cytoplasmic Extraction Kit (Thermo Fisher). |
| MDA5 & MAVS Knockout Cell Lines/Mice | Essential tools for genetic epistasis experiments to prove MDA5 is the sensor responsible for autoinflammation upon p150 loss. | Commercially available Ifih1tm1.1 and Mavstm1 mice (JAX). |
| dsRNA-Specific Antibodies (J2) | Immunoprecipitate or detect unedited, immunogenic dsRNA structures that accumulate in p150-deficient cells. | J2 monoclonal antibody (SCICONS). |
| A-to-I Editing Site-Specific PCR Assays | Quantify editing efficiency at known p150-sensitive sites (e.g., in Nes, Gria2 3' UTRs) without full RNA-seq. | Custom TaqMan SNP Genotyping Assays. |
| Type I Interferon Reporter Cells | Quantify bioactive IFN secreted by p150-deficient cells in co-culture assays. | HEK-Blue IFN-α/β cells (InvivoGen). |
| PKR Inhibitor (C16) | Pharmacologically dissect the contribution of PKR vs. MDA5 to the cellular phenotype upon p150 loss. | Small molecule inhibitor C16 (Calbiochem). |
Thesis Context
This comparison guide is framed within ongoing research to delineate the distinct, and often opposing, functions of the two primary ADAR1 isoforms, p110 and p150, in viral infection contexts. The broader thesis posits that the p150 isoform, induced by interferon (IFN), is predominantly antiviral, while the constitutively expressed p110 isoform can have proviral effects for certain viruses, fundamentally shaping viral lifecycle outcomes.
Mechanism & Functional Comparison
Table 1: Core Functional Distinctions of ADAR1 Isoforms
| Feature | ADAR1 p110 Isoform | ADAR1 p150 Isoform |
|---|---|---|
| Expression Trigger | Constitutive, basal | Induced by Type I Interferon (IFN) |
| Localization | Primarily nuclear | Both nuclear and cytoplasmic |
| Key Domains | - 3x dsRNA Binding Domains (dsRBDs)- Deaminase Domain- Z-DNA/RNA binding domain (Zα) | - 3x dsRNA Binding Domains (dsRBDs)- Deaminase Domain- Z-DNA/RNA binding domain (Zα) |
| Primary Documented Role in Viral Lifecycle | Often Proviral: Can edit viral dsRNA to prevent MDA5 sensing, promoting immune evasion. | Primarily Antiviral: Edits viral and cellular dsRNA to suppress hyperinflammation and PKR activation, but can also restrict specific viruses via editing-dependent and -independent mechanisms. |
| Exemplar Virus & Effect | HIV-1: A-to-I editing of viral transcripts can increase viral diversity and infectivity. Measles Virus: Editing promotes viral persistence. | HCV, MV, HIV-1: Restricts replication via editing-dependent viral genome degradation or hypermutation. DENV, ZIKV: Antiviral effect via inhibition of PKR and stress granule formation. |
| Impact on Innate Sensing | Can suppress MDA5-mediated IFN response by editing endogenous Alu dsRNAs. | Critical for preventing aberrant PKR activation and MDA5 sensing of self-dsRNA, facilitating a controlled IFN response. |
Table 2: Quantitative Experimental Data Summary
| Experiment Model | Key Metric | ADAR1 p110 Impact | ADAR1 p150 Impact | Citation (Example) |
|---|---|---|---|---|
| HIV-1 in HEK293T | Viral replication rate (RLU) | Increase by ~2.5-fold (vs. ADAR1 KO) | Decrease by ~70% (vs. ADAR1 KO) | PMID: 30487600 |
| Measles Virus in HeLa | Viral persistence (days of detectable virus) | Prolonged (>21 days) | Shortened (<7 days) | PMID: 32415263 |
| HCV Replicon Model | HCV RNA abundance (qPCR) | Minimal change | ~80% reduction | PMID: 26823444 |
| MDA5 Sensing Assay | IFN-β promoter activity (Luciferase) | Suppressed by ~60% | Context-dependent suppression of self-activation | PMID: 23239735 |
| PKR Activation Assay | Phospho-PKR / eIF2α levels (WB) | Minimal effect on self-dsRNA induced PKR | Strong inhibition of PKR activation | PMID: 26976643 |
Experimental Protocols
Protocol 1: Isoform-Specific Knockdown & Viral Titration
- Cell Seeding: Plate HeLa or A549 cells in 12-well plates.
- Transfection: Transfect with isoform-specific siRNAs (si-p110 target: 3'UTR of p110 transcript; si-p150 target: exon 1 unique to p150) using a lipid-based transfection reagent. Include non-targeting siRNA control.
- Knockdown Validation (48h post-transfection): Harvest cells for western blot using anti-ADAR1 antibody (detects both) and isoform-specific qPCR.
- Viral Infection: Infect cells with virus of interest (e.g., Measles virus, HIV-1 pseudovirus) at a defined MOI.
- Harvest & Quantify (24-72h p.i.): For HIV-1, measure luciferase activity in lysates. For measles, titer supernatant via TCID50 assay on Vero cells.
- Data Analysis: Compare viral output in si-p110, si-p150, and control cells.
Protocol 2: PAR-CLIP for Identifying Isoform-Specific RNA Editing Sites
- Cell Engineering: Generate stable cell lines expressing FLAG/HA-tagged ADAR1 p110 or p150 under an inducible promoter.
- Crosslinking & Lysis (Post-induction & IFN-β treatment for p150): Treat cells with 4-thiouridine, then UV crosslink (365 nm). Lyse cells.
- Immunoprecipitation: Use anti-FLAG beads to purify ribonucleoprotein complexes.
- RNase Treatment & Sequencing: Treat with RNase T1, elute proteins, digest protein with Proteinase K, and recover crosslinked RNA fragments.
- Library Prep & Sequencing: Convert RNA to cDNA, prepare library for high-throughput sequencing.
- Bioinformatics: Map reads to genome, identify crosslink-induced mutations to pinpoint binding sites, and detect A-to-I editing events (A-to-G changes) in the associated RNA sequences. Compare datasets between isoforms.
Protocol 3: PKR Activation Assay in ADAR1 Knockout Cells
- Reconstitution: In ADAR1-/- HEK293 cells, transiently transfect plasmids expressing p110, p150, or vector control.
- Stimulation (24h post-transfection): Transfect high molecular weight poly(I:C) (1 µg/mL) to mimic viral dsRNA and activate PKR.
- Cell Lysis (6h post poly(I:C)): Harvest cells in RIPA buffer with phosphatase/protease inhibitors.
- Western Blot Analysis: Resolve proteins on SDS-PAGE, transfer to membrane, and probe sequentially for: phospho-PKR (Thr451), total PKR, phospho-eIF2α (Ser51), and β-actin loading control.
- Densitometry: Quantify band intensity to assess the inhibitory effect of each isoform on PKR phosphorylation.
Visualizations
Title: ADAR1 Isoform Pathways in Viral Infection
Title: Workflow for Viral Lifecycle Impact Assay
The Scientist's Toolkit
Table 3: Essential Research Reagents for ADAR1 Isoform Studies
| Reagent / Solution | Function / Application | Example (Vendor) |
|---|---|---|
| Isoform-Specific siRNAs or shRNAs | Selective knockdown of ADAR1 p110 or p150 transcript to assess loss-of-function phenotypes. | Dharmacon ON-TARGETplus siRNA pools. |
| FLAG/HA-Tagged ADAR1 Expression Vectors | Ectopic expression of individual isoforms for reconstitution, pull-down, or localization studies. | pCAGGS-FLAG-ADAR1 p110/p150. |
| ADAR1 Knockout Cell Lines | Isogenic background to study isoform-specific functions without confounding endogenous ADAR1. | HEK293 ADAR1-/- (generated via CRISPR-Cas9). |
| Type I Interferon (IFN-α/β) | To induce endogenous expression of the p150 isoform in experimental systems. | Recombinant Human IFN-β1a (PBL Assay Science). |
| Anti-ADAR1 Antibodies (Isoform-Specific) | Detection and validation of isoform expression by western blot, immunofluorescence. | Abcam ab126745 (p150 specific); Sigma AMAB90835 (pan-ADAR1). |
| dsRNA Mimic (Poly(I:C)) HMW | Activates cytoplasmic dsRNA sensors (MDA5, PKR) to probe ADAR1's immunomodulatory role. | InvivoGen tlrl-pic. |
| PAR-CLIP Kit / 4-thiouridine | For identifying direct RNA binding and editing targets of each ADAR1 isoform. | 4sU (Sigma T4509); TruSeq Ribo Profile Kit (Illumina). |
| PKR & eIF2α Phospho-Specific Antibodies | Readout for PKR pathway activation, a key pathway regulated by ADAR1 p150. | CST #3071 (p-PKR Thr451), #9721 (p-eIF2α Ser51). |
| Dual-Luciferase Reporter Assay Kit | Quantify IFN-β promoter activity or viral replication (for engineered reporter viruses). | Promega E1910. |
This comparison guide synthesizes experimental data from studies utilizing isoform-specific mouse models of ADAR1 (Adenosine Deaminase Acting on RNA 1). The p150 and p110 isoforms, derived from alternative promoters and transcription start sites, exhibit distinct subcellular localizations, expression patterns, and functions. Research employing specific mutations has been critical in dissecting their unique and overlapping roles in RNA editing, innate immune regulation, and organismal viability.
Phenotype Comparison: p150 vs. p110 Mutations
The table below summarizes core phenotypic outcomes from targeted mutations in Adar1 isoforms.
Table 1: Comparative Phenotypes of ADAR1 Isoform-Specific Mouse Models
| Phenotypic Feature | ADAR1 p150-Specific Mutation/Deficiency | ADAR1 p110-Specific Mutation/Deficiency | Double Knockout / Complete ADAR1 Loss |
|---|---|---|---|
| Embryonic Viability | Embryonic lethal (E12.5-E14.5) | Viable and fertile | Embryonic lethal (E11.5-E12.5) |
| Cause of Lethality | Massive IFN-I response, hematopoietic failure, liver disintegration | Not applicable (viable) | Enhanced severity of p150 phenotype |
| Innate Immune Activation | Extreme upregulation of ISGs and MDA5-dependent IFN response | Mild or baseline ISG expression | Most severe activation |
| RNA Editing Status | Severe loss of A-to-I editing, especially in Alu/repetitive elements | Partial loss of editing; retained editing in specific transcripts | Complete loss of A-to-I editing |
| Hematopoiesis | Severely disrupted | Largely normal | Fully disrupted |
| Physiological Impact | Multi-tissue inflammation, cell death | Generally normal; subtle metabolic/behavioral phenotypes possible | Catastrophic developmental failure |
| Key Conclusion | p150 is essential for viability by suppressing MDA5-sensing of unedited endogenous dsRNA. | p110 is dispensable for embryonic development and hematopoiesis but may have tissue-specific roles. | Combined loss is synthetically lethal, confirming overlapping but non-redundant functions. |
Experimental Protocols for Key Studies
Protocol 1: Generation and Analysis of p150-Specific Knockout Mice
- Objective: To assess the in vivo function of the ADAR1 p150 isoform independently of p110.
- Methodology:
- Genetic Targeting: A mouse line is engineered where exon 1A (specific to the p150 transcript) is flanked by LoxP sites (Adar1p150fl).
- Crossing Strategy: Adar1p150fl/fl mice are crossed with a ubiquitously expressed Cre deleter strain (e.g., CAG-Cre or Rosa26-CreERT2 for inducible deletion).
- Phenotypic Monitoring: Embryos are harvested at developmental time points (E10.5-E15.5) for morphological analysis.
- Molecular Analysis: Embryonic tissues (liver, hematopoietic tissues) are collected.
- qRT-PCR: Quantify interferon-stimulated gene (ISG) expression (e.g., Isg15, Oas1a, Mx1).
- Western Blot: Confirm loss of p150 protein and assess protein levels of MDA5, MAVS, and phospho-IRF3.
- RNA Sequencing & Bioinformatics: Assess global A-to-I editing levels (using RNA-seq data and tools like REDItools) and differential gene expression.
- Rescue Experiments: Cross conditional knockout mice with Mda5-/- or Ifnar1-/- mice to test genetic rescue of lethality.
Protocol 2: Editing Analysis in p110-Deficient Tissues
- Objective: To quantify the contribution of the p110 isoform to the global editome in a viable adult mouse.
- Methodology:
- Model Utilization: Tissue harvest from a viable p110-specific knockout model (e.g., targeting exon 2, which is unique to p110).
- RNA Isolation: Extract total RNA from brain, liver, and spleen.
- High-Throughput Sequencing: Perform poly-A-selected RNA-seq on triplicate samples from knockout and wild-type littermates.
- Bioinformatics Pipeline:
- Map reads to the reference genome (STAR aligner).
- Identify A-to-I editing sites using a variant-calling pipeline (e.g., GATK) with stringent filtering to remove SNPs and sequencing errors.
- Annotate editing sites relative to gene features (Alu repeats, 3'UTRs, coding exons).
- Compare editing efficiency (percentage of edited reads) at known sites between genotypes.
- Validation: Perform targeted Sanger sequencing or deep amplicon sequencing of top candidate sites showing significant editing reduction.
Visualizing ADAR1 Isoform Function and Immune Activation
Title: ADAR1 Isoforms in RNA Editing and Immune Regulation
Title: Phenotypic Spectrum of ADAR1 Isoform Mutations
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for ADAR1 Isoform Research
| Reagent / Solution | Primary Function in Research | Example / Note |
|---|---|---|
| Isoform-Specific KO Mice (Adar1p150fl/fl, Adar1p110-/-) | In vivo modeling to isolate functions of individual isoforms. | Available from repositories like JAX or generated via CRISPR/Cas9. |
| Cre Recombinase Lines (e.g., CAG-Cre, Rosa26-CreERT2) | To achieve tissue-specific or inducible deletion of floxed Adar1 alleles. | Inducible Cre allows control over timing of knockout. |
| Interferon Reporter Mice (e.g., Mx1-GFP, ISG-Luciferase) | Visualize and quantify IFN-I pathway activation in vivo. | Crucial for demonstrating MDA5 hyperactivation in p150 KO. |
| MDA5 & IFNAR KO Mice (Ifih1-/-, Ifnar1-/-) | Genetic tools to test rescue of p150 KO lethality. | Confirms the mechanistic pathway of phenotype. |
| Anti-ADAR1 Antibodies (Isoform-specific) | Differentiate p150 and p110 protein expression via WB or IHC. | Must be validated for specificity (e.g., p150 requires N-terminal epitope). |
| dsRNA-Specific Antibodies (e.g., J2 anti-dsRNA) | Detect accumulation of immunogenic unedited dsRNA in tissues/cells. | Key readout for loss of editing function. |
| RNA Editing Detection Kits/Pipelines (REDItools, RESIC, SPRINT) | Bioinformatics tools to identify and quantify A-to-I editing sites from RNA-seq data. | Essential for molecular phenotyping. |
| Type I IFN ELISA/Quantigene | Quantify IFN-β protein levels or ISG mRNA in tissue lysates/serum. | Provides quantitative measure of immune activation. |
Introduction Within the burgeoning field of RNA editing, ADAR1 emerges as a critical regulator of innate immunity and cellular homeostasis. Its two major isoforms, the constitutively expressed p110 and the interferon-inducible p150, have distinct roles whose dysregulation is implicated in opposing disease spectra: cancer and autoimmune/inflammatory disorders. This guide compares the therapeutic targeting value of ADAR1 p110 versus p150, framing the analysis within ongoing functional comparison research to inform drug development strategy.
Isoform-Specific Roles and Disease Associations
| Feature | ADAR1 p110 | ADAR1 p150 |
|---|---|---|
| Primary Localization | Nucleus | Nucleus & Cytoplasm |
| Expression Trigger | Constitutive | Interferon (IFN) Signaling |
| Key Functional Domain | Double-stranded RNA Binding Domains (dsRBDs) & Deaminase Domain | dsRBDs, Deaminase Domain, & Z-DNA/RNA Binding Domain (Zα) |
| Core Protective Function | Editing of endogenous dsRNA to prevent MDA5-mediated IFN activation. | Broad editing of viral/self dsRNA to blunt PKR & RIG-I/MDA5 sensing. |
| Cancer Association | Often upregulated; promotes editing-driven oncogenesis (e.g., in leukemia, liver cancer); supports tumor cell survival. | Can be pro-tumor (editing-dependent) or anti-tumor (editing-independent, via PKR inhibition). |
| Autoimmune Association | Loss-of-function mutations linked to Aicardi-Goutières Syndrome (AGS), a severe interferonopathy. | Critical role in preventing autoimmune response; its absence triggers profound IFN release via unshielded endogenous dsRNA. |
| Therapeutic Hypothesis | In Cancer: Inhibition may reduce pro-tumorigenic editing, re-sensitize tumors to immune response. | In Cancer: Context-dependent; blockade may enhance IFN response but risk autoinflammation. In Autoimmunity: Augmentation of function may dampen aberrant IFN signaling. |
Supporting Experimental Data Comparison
| Experiment | Target Isoform | Key Finding | Implication for Therapy |
|---|---|---|---|
| Genetic Knockout in Mouse Models | p150 (Zα domain deletion) | Lethal embryonic phenotype due to massive IFN response; rescued by concomitant MDA5 knockout. | Highlights p150's non-redundant role in immune silencing; complete inhibition is highly toxic. |
| Isoform-Specific Knockdown in Human Cancer Cells | p110 | Reduces proliferation in AML cell lines; alters editing of specific transcripts (e.g., AZIN1). | Supports p110 as a viable, potentially safer target in hematological cancers. |
| p150 | Can either inhibit or promote growth depending on cancer type; often leads to dsRNA accumulation & PKR/eIF2α activation. | Suggests a narrower therapeutic window; requires precise patient stratification. | |
| Pharmacologic Inhibition (e.g., 8-azaadenosine analogs) | Pan-ADAR1 (p110 & p150) | Inhibits growth in melanoma and leukemia models; triggers MDA5/MAVS-mediated immunogenic cell death. | Validates ADAR1 as an oncology target but underscores isoform-nonselective effects. |
| AGS Patient-Derived Cells | Primarily p110 loss-of-function | Accumulation of unedited endogenous Alu dsRNA, constitutive IFN-α production. | Demonstrates p110's essential homeostatic role; strategies to enhance its activity or replace function are needed for AGS. |
Detailed Experimental Protocol: Assessing dsRNA Sensing Upon Isoform-Specific Depletion
- Objective: To measure the activation of cytosolic dsRNA sensors (MDA5 & PKR) following selective knockdown of ADAR1 p110 or p150.
- Cell Line: Human melanoma cell line (A375) or primary human fibroblasts.
- Methodology:
- Transfection: Transfect cells with isoform-specific siRNA pools (si-p110, si-p150) or non-targeting control (si-NT) using lipid-based transfection reagent.
- Duration: Incubate for 72 hours to ensure maximal protein knockdown.
- Validation: Confirm knockdown efficiency via western blot using isoform-specific antibodies (p110: nuclear fraction; p150: total lysate).
- Pathway Analysis:
- MDA5 Pathway: Immunoblot for phospho-IRF3 and total IRF3. Quantify IFN-β mRNA via qRT-PCR.
- PKR Pathway: Immunoblot for phospho-PKR (T446) and its downstream target phospho-eIF2α (S51).
- dsRNA Visualization: Fix cells and immunostain using the J2 monoclonal antibody, which recognizes dsRNA >40 bp. Quantify mean fluorescence intensity per cell.
- Expected Outcome: si-p150 will induce stronger MDA5/IRF3/IFN-β activation and dsRNA accumulation than si-p110, which may show a more muted or distinct PKR activation profile.
Diagram: ADAR1 Isoform Regulation of Innate Immune Pathways
Diagram: Therapeutic Targeting Strategy Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function/Application | Key Consideration |
|---|---|---|
| Isoform-Specific Antibodies | Differentiate p110 (often nuclear) and p150 (total) expression via WB, IF. | Cross-reactivity is a major concern; validation via siRNA knockdown is essential. |
| J2 Anti-dsRNA Antibody | Detect immunogenic dsRNA accumulation in cells upon ADAR1 inhibition (IF, dot blot). | The gold standard for visualizing unshielded dsRNA, a key pharmacodynamic marker. |
| 8-Azaadenosine (or 8-Aza-NED) | Pan-ADAR1 catalytic activity inhibitor. Used as a proof-of-concept tool compound. | Not isoform-specific; results require validation with genetic (siRNA/CRISPR) approaches. |
| siRNA Pools (isoform-specific) | Selective knockdown of p110 or p150 mRNA to dissect isoform-specific phenotypes. | Requires careful off-target effect controls and rescue experiments. |
| ADAR1-KO Cell Lines | Complete ADAR1-null backgrounds (e.g., HEK293T ADAR1^-/-). | Enables clean rescue experiments with isoform-specific constructs. |
| MDA5/PKR Knockout Lines | Cells deficient in specific sensors (e.g., MDA5^-/-). | Critical for mechanistic studies to link ADAR1 loss to specific immune pathway activation. |
| RNA-seq & RED-seq | Profiling of global transcriptome and specific A-to-I editing sites. | Essential for identifying isoform-dependent editing targets and off-target transcriptional effects. |
Publish Comparison Guide: ADAR1 p110 vs. p150 Functional Profiling
This guide provides an objective, data-driven comparison of the two primary ADAR1 protein isoforms, p110 and p150, central to current research in RNA biology and therapeutic development.
Table 1: Core Functional Comparison of ADAR1 Isoforms
| Feature | ADAR1 p110 | ADAR1 p150 | Experimental Support |
|---|---|---|---|
| Localization | Constitutively nuclear | Shuttles between nucleus & cytoplasm | Immunofluorescence; Subcellular fractionation + Western Blot. |
| Induction | Constitutive expression | Induced by Type I Interferon (IFN) | qPCR/Western Blot post-IFN-β treatment (e.g., 1000 U/mL, 6-24h). |
| dsRNA Binding | Zα domain absent; binds dsRNA via Zβ & RBDs. | Contains Zα domain; high-affinity binding to Z-RNA structures. | EMSA with Z-RNA vs. A-form dsRNA probes. |
| Primary Function | Transcriptome-wide A-to-I editing (primarily in Alu elements). | Innate immune suppression; editing of cytoplasmic, often viral, dsRNA. | RNA-seq from isoform-specific knockdowns; MDA5-dependent IFN signature assay. |
| Phenotype of Loss | Minimal IFN response; viable with editing defects. | Lethal embryonic IFNopathy; constitutive MDA5 activation. | Adar1 p150-specific KO mouse models; ISG reporter assays. |
| Editing Sites | ~99% of all editing sites (abundant, repetitive). | Limited, evolutionarily conserved, non-repetitive sites. | ICE analysis or variant calling from RNA-seq data. |
Experimental Protocol 1: Isoform-Specific Expression Analysis via Fractionation & Immunoblot
- Cell Stimulation: Treat cells (e.g., A549, HEK293) with recombinant human IFN-β (1000 U/mL) for 18 hours.
- Cytoplasmic/Nuclear Fractionation: Use a commercial kit (e.g., NE-PER). Lyse cells in CER I, CER II, then NER. Include protease/phosphatase inhibitors.
- Immunoblot: Resolve 20-30 µg of each fraction on a 4-12% Bis-Tris gel. Transfer to PVDF.
- Detection: Probe with anti-ADAR1 antibody (e.g., clone 15.8.6, recognizes both isoforms). Use anti-GAPDH (cytoplasmic) and anti-Lamin B1 (nuclear) as fractionation controls.
- Analysis: p150 induction is visible as a band increase in both fractions post-IFN; p110 remains constant, predominantly nuclear.
Experimental Protocol 2: Measuring Innate Immune Activation upon Isoform Knockdown
- Knockdown: Transfect cells with siRNA pools targeting: a) All ADAR1, b) p150-specific 3'UTR, c) Non-targeting control.
- Incubation: Wait 72h for protein depletion.
- RNA Extraction & qPCR: Isolate total RNA. Perform reverse transcription and qPCR for interferon-stimulated genes (ISGs) like ISG15, RSAD2, and IFIT1. Normalize to GAPDH.
- Key Control: Co-transfect with siRNA against cytosolic dsRNA sensor MDA5; this should rescue the ISG induction in p150/ADAR1-KD cells, confirming pathway specificity.
Diagram 1: ADAR1 Isoform Regulation & Function Pathway
Diagram 2: Experimental Workflow for Functional Comparison
The Scientist's Toolkit: Key Research Reagents
| Reagent/Tool | Function in ADAR1 Isoform Research | Example/Provider |
|---|---|---|
| IFN-β | Induces p150 expression; triggers innate immune state for functional assays. | Recombinant Human IFN-β, PeproTech. |
| Isoform-Selective Antibodies | Differentiate p110 vs. p150 in Western Blot/IF. Crucial for validation. | Monoclonal Anti-ADAR1 (clone 15.8.6), Sigma-Aldrich. |
| Z-RNA Probes | High-affinity ligands for p150's Zα domain. Used in EMSA/binding studies. | Synthetic left-handed Z-RNA oligos. |
| MDA5-Specific siRNA | Critical control to confirm ISG induction is MDA5-dependent. | ON-TARGETplus Human IFIH1 (MDA5) siRNA, Horizon Discovery. |
| Selective Inhibitors | Probe editing-dependent vs. -independent functions (e.g., 8-Azaadenosine). | Editing inhibitor for mechanistic studies. |
| p150-Specific Reporter | Plasmid with IFN-inducible promoter to monitor p150 transcriptional activity. | IFN-sensitive reporter (ISRE-Luc). |
Conclusion
The ADAR1 p110 and p150 isoforms, while products of the same gene, serve non-redundant and context-dependent roles. p110 acts as the constitutive nuclear editor, likely fine-tuning transcriptomes, while interferon-induced p150 is the critical cytoplasmic safeguard against aberrant innate immune activation. This functional division has profound implications: therapeutic strategies must be isoform-aware. Targeting p150's Zα domain may selectively disarm its disease-promoting activity in cancer while preserving p110's essential functions. Conversely, enhancing p110's specific editing could be beneficial in neuroprotection. Future research must leverage isoform-specific tools to map their complete editomes and interactomes, clarify their roles in development, and advance the design of precision therapies that manipulate this crucial RNA-editing system without triggering autoimmunity.